How many \[\sigma \] and \[\pi \] – bonds are present in peroxymonosulfuric acid?
Answer
616.5k+ views
Hint: So, for finding sigma and pi bonds we have to draw the bond-line structure of peroxymonosulfuric acid and then count each and every bond between any of two atoms or elements.
Complete step by step solution:
Sigma bonds (σ bonds) are the strongest type of covalent chemical bonds. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups.
We know that Pi bond (\[\pi \]bond) is a bond formed by the overlap of p-orbitals on adjacent atoms, perpendicular to any sigma bond(s) between the same atoms. It is indicated in a Kekule structure or bond-line structure as an extra line parallel to the line which represents the sigma bond.
Chemical formula of peroxymonosulfuric acid is: \[{{H}_{2}}S{{O}_{5}}\]
And structure of peroxymonosulfuric acid:
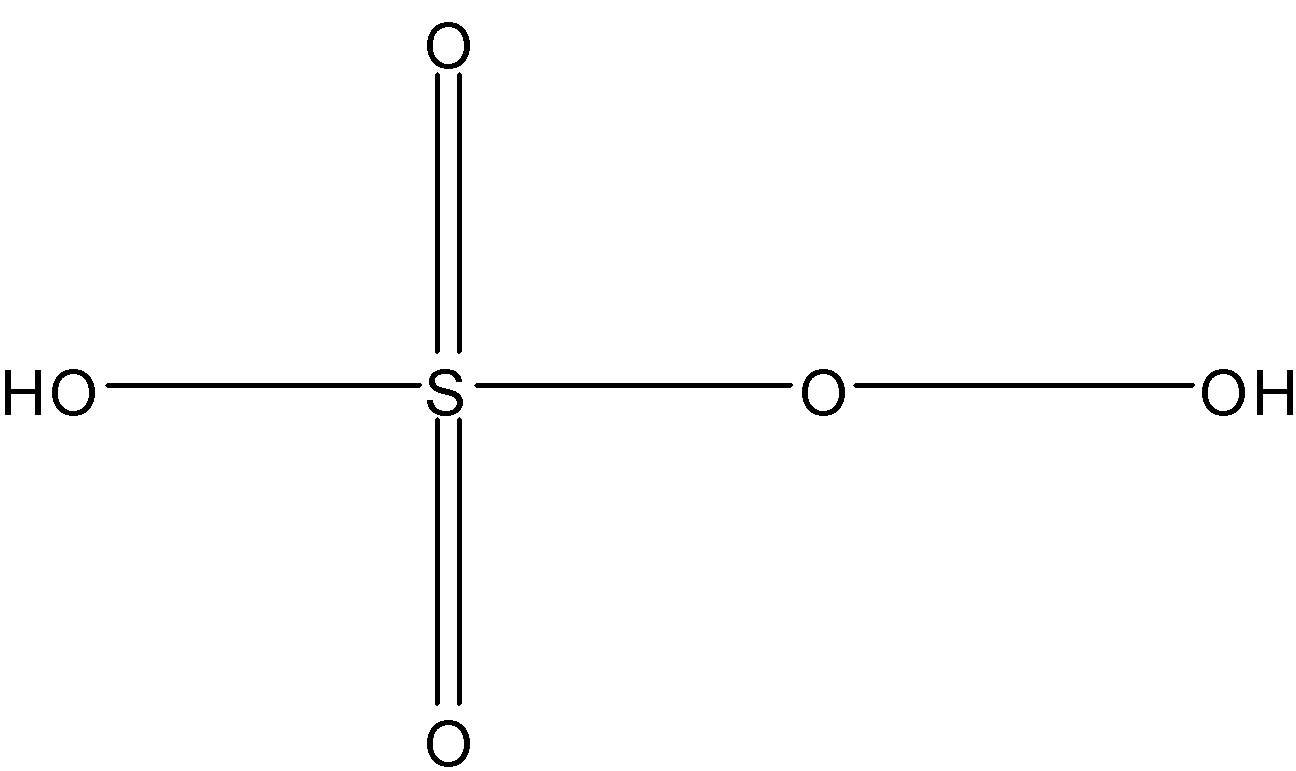
Here, number of sigma bonds = 4 sigma bonds between S and O + 2 sigma bonds between H and O + 1 sigma bond between O and O
= 7
Number of pi bonds = there are two pi bonds between S and O = 2
So, the answer is “7, 2”.
Note: If there is a double or triple bond between any of two atoms then 1st bond we count as sigma bond and other two or one bond we count as pi bond. And in this compound a peroxy bond (O-O).
Complete step by step solution:
Sigma bonds (σ bonds) are the strongest type of covalent chemical bonds. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups.
We know that Pi bond (\[\pi \]bond) is a bond formed by the overlap of p-orbitals on adjacent atoms, perpendicular to any sigma bond(s) between the same atoms. It is indicated in a Kekule structure or bond-line structure as an extra line parallel to the line which represents the sigma bond.
Chemical formula of peroxymonosulfuric acid is: \[{{H}_{2}}S{{O}_{5}}\]
And structure of peroxymonosulfuric acid:

Here, number of sigma bonds = 4 sigma bonds between S and O + 2 sigma bonds between H and O + 1 sigma bond between O and O
= 7
Number of pi bonds = there are two pi bonds between S and O = 2
So, the answer is “7, 2”.
Note: If there is a double or triple bond between any of two atoms then 1st bond we count as sigma bond and other two or one bond we count as pi bond. And in this compound a peroxy bond (O-O).
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




