Show on a graph, the variation of resistivity with temperature for a typical semiconductor.
Answer
617.7k+ views
Hint: In semiconductors, the forbidden gap between the conduction band and the valence bad is exceedingly small.
Complete step by step solution:
For a semiconductor, resistivity decreases rapidly with increase in temperature. Same current will flow through all the resistors.
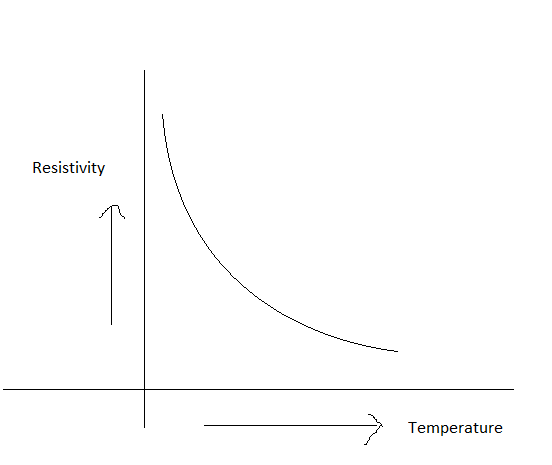
The above graph shows the variation of resistivity with temperature for a typical semiconductor.
Additional information: Resistivity is known as specific electrical resistance or volume resistivity. It is defined as the intrinsic property of a given material that shows how it opposes the flow of current. In other words, it can also define the resistance offered by the conductor having unit length and unit area of cross section. So, it does not depend upon the length and area of the cross section of a material. But the resistance of a material depends upon the length and area of the cross section of the material.
Silicon is a semiconductor. In semiconductors, the forbidden gap between the conduction band and the valence bad is exceedingly small. At 0 kelvin, the valence band is completely filled, and the conduction band may be empty. But when a small amount of energy is applied, the electrons easily move to the conduction band. Silicon is therefore a good example for semiconductors. Under normal circumstances silicon acts as a poor conductor. Each silicon atom is bonded to 4 other silicon atoms. The bonds between these atoms are covalent bonds where the electrons are in fixed positions.
Note: Before solving the question students need to be well versed with all the properties of semiconductors and also how they change under different circumstances for example pressure and temperature.
Complete step by step solution:
For a semiconductor, resistivity decreases rapidly with increase in temperature. Same current will flow through all the resistors.

The above graph shows the variation of resistivity with temperature for a typical semiconductor.
Additional information: Resistivity is known as specific electrical resistance or volume resistivity. It is defined as the intrinsic property of a given material that shows how it opposes the flow of current. In other words, it can also define the resistance offered by the conductor having unit length and unit area of cross section. So, it does not depend upon the length and area of the cross section of a material. But the resistance of a material depends upon the length and area of the cross section of the material.
Silicon is a semiconductor. In semiconductors, the forbidden gap between the conduction band and the valence bad is exceedingly small. At 0 kelvin, the valence band is completely filled, and the conduction band may be empty. But when a small amount of energy is applied, the electrons easily move to the conduction band. Silicon is therefore a good example for semiconductors. Under normal circumstances silicon acts as a poor conductor. Each silicon atom is bonded to 4 other silicon atoms. The bonds between these atoms are covalent bonds where the electrons are in fixed positions.
Note: Before solving the question students need to be well versed with all the properties of semiconductors and also how they change under different circumstances for example pressure and temperature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

The highest temperature in Karnataka is recorded in class 10 social science CBSE




