What is the reaction of epoxide with Grignard reagent?
Answer
559.5k+ views
Hint: Grignard reagents are organometallic compounds having a metal and an organic group along with a halogen, with the formula, RMgX, where R is any alkyl group, Mg is magnesium and X is halogen. They undergo additional reactions with carbonyl groups. Epoxides are tricyclic compounds having one carbonyl group.
Complete answer:
Grignard reagents are highly basic organometallic compounds. They have the tendency to react with any species that donates a proton. Grignard reagents are capable of generating a nucleophile, which can react with electrophiles in addition to reactions. On the other hand, epoxides are tricyclic compounds having 2 carbon and 1 oxygen atoms. They, when undergoes addition with Grignard reagents, leads to the ring opening of the epoxides.
The reaction of epoxide with Grignard reagents leads to the formation of primary alcohols, with two carbons more than the length, due to the ring opening. The reaction of epoxide with Grignard reagents is:
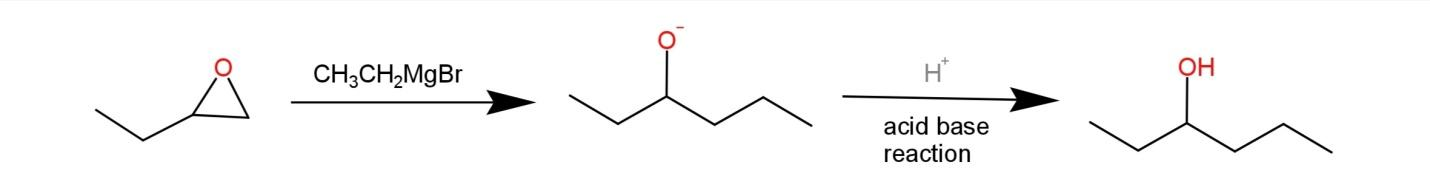
Epoxide opens as the electrophile is generated by the addition of the alkyl group by Grignard reagent on epoxide. This when subjected to acidic conditions leads to the formation of a primary alcohol.
Hence, reaction of epoxides with Grignard reagent generates primary alcohols.
Note:
Epoxides are also called ethylene oxides. The reaction follows the mechanism of nucleophilic substitution, which is ${{S}_{N}}^{2}$ type. The strong basic condition of the Grignard reagent is responsible for the opening of the ring of the epoxides. The acid workup is the final step in the reaction of epoxides. Apart from reaction with epoxides, Grignard also reacts with aldehydes, ketones, acid chlorides, etc. compounds with a carbonyl group.
Complete answer:
Grignard reagents are highly basic organometallic compounds. They have the tendency to react with any species that donates a proton. Grignard reagents are capable of generating a nucleophile, which can react with electrophiles in addition to reactions. On the other hand, epoxides are tricyclic compounds having 2 carbon and 1 oxygen atoms. They, when undergoes addition with Grignard reagents, leads to the ring opening of the epoxides.
The reaction of epoxide with Grignard reagents leads to the formation of primary alcohols, with two carbons more than the length, due to the ring opening. The reaction of epoxide with Grignard reagents is:

Epoxide opens as the electrophile is generated by the addition of the alkyl group by Grignard reagent on epoxide. This when subjected to acidic conditions leads to the formation of a primary alcohol.
Hence, reaction of epoxides with Grignard reagent generates primary alcohols.
Note:
Epoxides are also called ethylene oxides. The reaction follows the mechanism of nucleophilic substitution, which is ${{S}_{N}}^{2}$ type. The strong basic condition of the Grignard reagent is responsible for the opening of the ring of the epoxides. The acid workup is the final step in the reaction of epoxides. Apart from reaction with epoxides, Grignard also reacts with aldehydes, ketones, acid chlorides, etc. compounds with a carbonyl group.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




