Reaction of aniline with \[{\text{HN}}{{\text{O}}_{\text{2}}}\] followed by treatment of dilute acid gives:
A) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHOH}}\]
B) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{OH}}\]
C) \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{NHN}}{{\text{H}}_{\text{2}}}\]
D) \[{{\text{C}}_{\text{6}}}{{\text{H}}_6}\]
Answer
597.6k+ views
Hint:Aniline reacts similarly to primary aliphatic amines. Aniline gives diazonium ion after treatment with nitrous acid. On further treatment with dilute acid undergo hydrolysis reaction.
Complete solution:
In this reaction, aniline is the reactant and reagent given to us is nitrous acid, \[{\text{HN}}{{\text{O}}_{\text{2}}}\] and dilute acid.
Aniline is the simplest aromatic amine that behaves similarly to primary aliphatic amines. It consists of an amino group bonded to a phenyl group.
Reagent nitrous acid is prepared by reaction of sodium nitrite with hydrochloric acid. It is a weak acid
\[{\text{NaN}}{{\text{O}}_{\text{2}}}{\text{ + HCl}} \to {\text{ NaCl + HN}}{{\text{O}}_{\text{2}}}\]
Now, we will see the reaction mechanism when of aniline reacts with \[{\text{HN}}{{\text{O}}_{\text{2}}}\] followed by treatment with dilute acid.
Step 1: Conversion of aniline to diazonium ion using nitrous acid, \[{\text{HN}}{{\text{O}}_{\text{2}}}\]reagent.
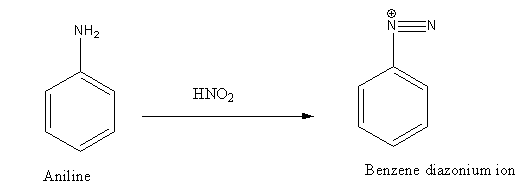
Step 2: Diazonium ion on further treatment with dilute acid undergoes hydrolysis reaction and gives phenol as the product.
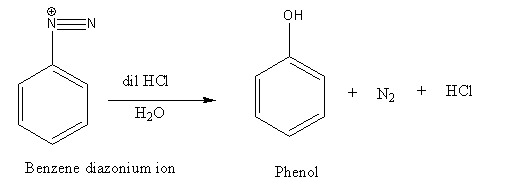
Thus, the correct options are (B).
Note:Aromatic diazonium ion is a very important intermediate to produce substituted benzene. The final product depends on reagent use. Here the reagent used is dilute acid so the diazonium ion undergoes a hydrolysis reaction. The addition of water molecules is known as the hydrolysis reaction. Amines on hydrolysis give alcohol as the product while aniline gives phenol as the product.
Complete solution:
In this reaction, aniline is the reactant and reagent given to us is nitrous acid, \[{\text{HN}}{{\text{O}}_{\text{2}}}\] and dilute acid.
Aniline is the simplest aromatic amine that behaves similarly to primary aliphatic amines. It consists of an amino group bonded to a phenyl group.
Reagent nitrous acid is prepared by reaction of sodium nitrite with hydrochloric acid. It is a weak acid
\[{\text{NaN}}{{\text{O}}_{\text{2}}}{\text{ + HCl}} \to {\text{ NaCl + HN}}{{\text{O}}_{\text{2}}}\]
Now, we will see the reaction mechanism when of aniline reacts with \[{\text{HN}}{{\text{O}}_{\text{2}}}\] followed by treatment with dilute acid.
Step 1: Conversion of aniline to diazonium ion using nitrous acid, \[{\text{HN}}{{\text{O}}_{\text{2}}}\]reagent.

Step 2: Diazonium ion on further treatment with dilute acid undergoes hydrolysis reaction and gives phenol as the product.

Thus, the correct options are (B).
Note:Aromatic diazonium ion is a very important intermediate to produce substituted benzene. The final product depends on reagent use. Here the reagent used is dilute acid so the diazonium ion undergoes a hydrolysis reaction. The addition of water molecules is known as the hydrolysis reaction. Amines on hydrolysis give alcohol as the product while aniline gives phenol as the product.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What is the Full Form of PVC, PET, HDPE, LDPE, PP and PS ?

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

The electric field due to a uniformly charged nonconducting class 12 physics CBSE

In a halfwave rectifier the rms value of the ac component class 12 physics CBSE

What is apomixis and what is its importance class 12 biology CBSE




