How many pairs of electrons do the two oxygen atoms in an oxygen molecule share with each other?
Answer
562.5k+ views
Hint: When a pair of electrons are shared by two atoms, a covalent bond is formed. So to determine the pair of electrons shared by the two oxygen atoms, we need to find the bonding in the oxygen molecule.
Complete answer:
To determine the bonding in an oxygen molecule, we first need to know its molecular formula. The molecular formula of oxygen molecules is ${{O}_{2}}$.
Now, the chemical bonding in a molecule can be depicted using the Lewis dot structures.
To draw the Lewis dot structure of oxygen molecule,
- We first need to know the electronic configuration of oxygen atoms.
\[^{8}O=1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\]
- Determine the number of valence electrons.
Oxygen atom has 6 electrons in its outermost (n = 2) shell and hence it has 6 valence electrons.
The valence electrons are the ones that participate in molecule or chemical bond formation.
- Arrange the valence electrons around the oxygen atoms.
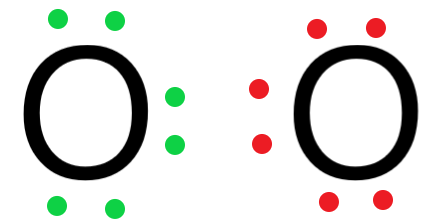
- Since the oxygen atom has 6 valence electrons, it needs 2 electrons to completely fill its outermost shell and satisfy the octet rule.
The two electrons required by the oxygen atom can be obtained by the other oxygen molecule by sharing.
Each oxygen atom will share two electrons with the other oxygen atom and form two covalent bonds or a double bond.
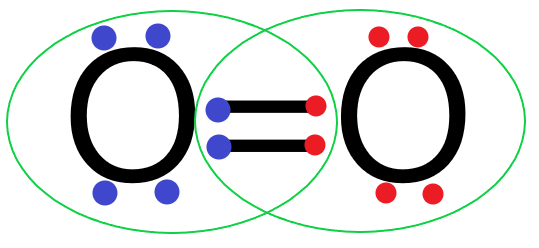
So we can see that the two oxygen atoms in an oxygen molecule share 2 pairs of electrons with each other
Note:
When there exists a charge atom, electrons equal to the magnitude of the charge are added (in case of a negatively charged anion) or subtracted (in case of a positive charged cation) from the Lewis dot diagram.
Complete answer:
To determine the bonding in an oxygen molecule, we first need to know its molecular formula. The molecular formula of oxygen molecules is ${{O}_{2}}$.
Now, the chemical bonding in a molecule can be depicted using the Lewis dot structures.
To draw the Lewis dot structure of oxygen molecule,
- We first need to know the electronic configuration of oxygen atoms.
\[^{8}O=1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\]
- Determine the number of valence electrons.
Oxygen atom has 6 electrons in its outermost (n = 2) shell and hence it has 6 valence electrons.
The valence electrons are the ones that participate in molecule or chemical bond formation.
- Arrange the valence electrons around the oxygen atoms.

- Since the oxygen atom has 6 valence electrons, it needs 2 electrons to completely fill its outermost shell and satisfy the octet rule.
The two electrons required by the oxygen atom can be obtained by the other oxygen molecule by sharing.
Each oxygen atom will share two electrons with the other oxygen atom and form two covalent bonds or a double bond.

So we can see that the two oxygen atoms in an oxygen molecule share 2 pairs of electrons with each other
Note:
When there exists a charge atom, electrons equal to the magnitude of the charge are added (in case of a negatively charged anion) or subtracted (in case of a positive charged cation) from the Lewis dot diagram.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




