Oxidation state of S in \[{H_2}{S_2}{O_8}\] is:
(A) +6
(B) +7
(C) +8
(D) 0
Answer
558.1k+ views
Hint: The name of the compound given in the question is Persulfuric acid. It contains peroxide linkage. The oxidation state of the oxygen atoms differ in case of peroxides from the normal oxidation state of the oxygen atoms.
Complete answer: The name of the compound given in the question is Persulfuric acid. As the name suggests, the compound has a peroxide group. Let’s see the chemical structure of \[{H_2}{S_2}{O_8}\] in order to find oxidation number of S.
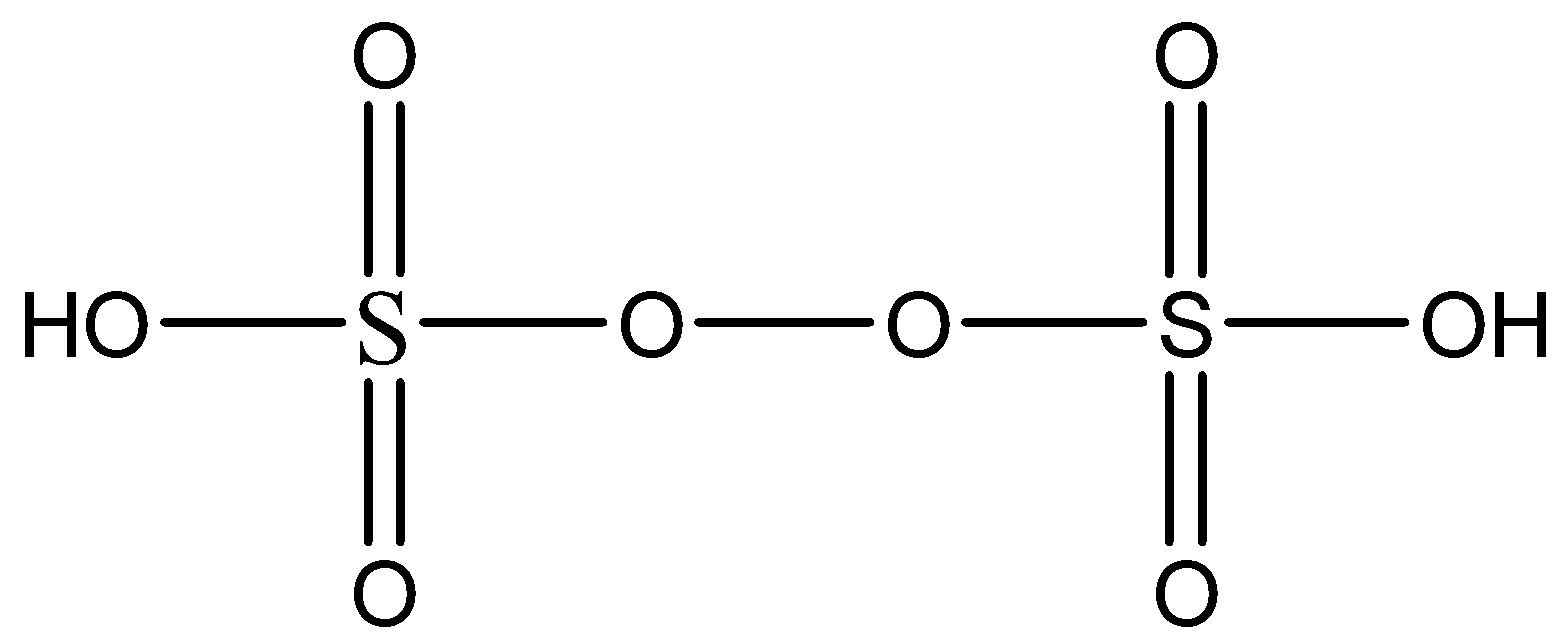
Now we know that we can find the oxidation number of any atom in a species by a fact that simply adding the oxidation numbers of all the atoms will give the overall charge on the species.
- Remember that both the oxygen atoms in the peroxide group are given an oxidation number of (-1). This is because each of this oxygen is bonded to another oxygen atom hence its oxidation number increases to (-1).
So, we can write that overall charge on \[{H_2}{S_2}{O_8}\] = 2(Oxidation number of S atom) + 2(Oxidation number of H atom) + 6(Oxidation number of normal oxygen atom) + 2(Oxidation number of peroxide oxygen)
We have multiplied the oxidation number of atoms according to the number of atoms present in the compound.
So, 0 = 2(Oxidation number of S atom) + 2(+1) + 6(-2) + 2(-1)
0 = 2(Oxidation number of S atom) + 2 - 12 -2
0 = 2(Oxidation number of S atom) - 12
2(Oxidation number of S atom) = 12
Oxidation number of S atom = +6
So, the correct answer is “Option A”.
Note:Do not consider that all the oxygen atoms have (-2) oxidation state in this compound. Even in the process of finding oxidation states of atoms in other compounds, make sure that you check the form of oxygen atoms, as they have different oxidation states if they are present as peroxide, superoxide or normal form.
Complete answer: The name of the compound given in the question is Persulfuric acid. As the name suggests, the compound has a peroxide group. Let’s see the chemical structure of \[{H_2}{S_2}{O_8}\] in order to find oxidation number of S.

Now we know that we can find the oxidation number of any atom in a species by a fact that simply adding the oxidation numbers of all the atoms will give the overall charge on the species.
- Remember that both the oxygen atoms in the peroxide group are given an oxidation number of (-1). This is because each of this oxygen is bonded to another oxygen atom hence its oxidation number increases to (-1).
So, we can write that overall charge on \[{H_2}{S_2}{O_8}\] = 2(Oxidation number of S atom) + 2(Oxidation number of H atom) + 6(Oxidation number of normal oxygen atom) + 2(Oxidation number of peroxide oxygen)
We have multiplied the oxidation number of atoms according to the number of atoms present in the compound.
So, 0 = 2(Oxidation number of S atom) + 2(+1) + 6(-2) + 2(-1)
0 = 2(Oxidation number of S atom) + 2 - 12 -2
0 = 2(Oxidation number of S atom) - 12
2(Oxidation number of S atom) = 12
Oxidation number of S atom = +6
So, the correct answer is “Option A”.
Note:Do not consider that all the oxygen atoms have (-2) oxidation state in this compound. Even in the process of finding oxidation states of atoms in other compounds, make sure that you check the form of oxygen atoms, as they have different oxidation states if they are present as peroxide, superoxide or normal form.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




