One end of a copper rod of uniform cross-section and length 3.1 m is kept in contact with ice and the other end with water at ${{100}^{\circ }}C$. At what point along its length should a temperature of ${{200}^{\circ }}C$ be maintained so that in steady-state, the mass of ice melting be equal to that of the steam produced in the same interval of time? Assume that the whole system is insulated from the surrounding. Latent heat of fusion of ice and vaporization of water is 80 cal/gm and 540 cal/gm respectively
a) 40 cm from ${{100}^{\circ }}C$
b) 40 cm from ${{0}^{\circ }}C$
c) 125 cm from ${{100}^{\circ }}C$
d) 125 cm from ${{0}^{\circ }}C$
Answer
601.8k+ views
Hint: Let us assume that the point will lie at distance x m. Now, by using the formula $Q=\dfrac{KA}{l}\Delta T$ , find the rate of heat for both ice and water. Also, we know that: $Q=m\times s$, where m is mass and s is the specific heat of fusion for ice and specific heat of evaporation for water. So, get the value of specific heats in terms of x. Divide both the equations and substitute the value of latent heats and get the value of x.
Complete step by step answer:
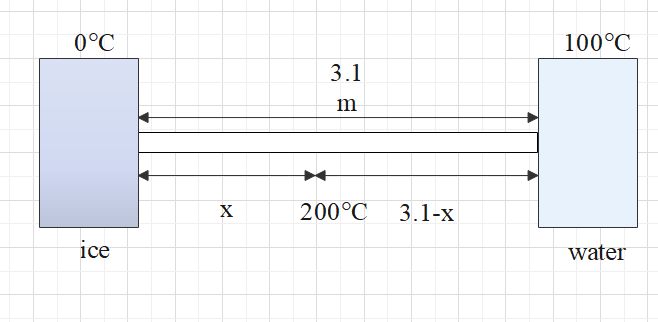
The heat rate by using the formula $Q=\dfrac{KA}{l}\Delta T=m\times s$
For ice:
\[{{Q}_{1}}=\dfrac{KA}{x}\left( 200-0 \right)=m\times {{s}_{ice}}......(1)\]
For water:
${{Q}_{2}}=\dfrac{KA}{\left( 3.1-x \right)}\left( 200-100 \right)=m\times {{s}_{water}}......(2)$
Divide equation (1) by (2), we get:
\[\begin{align}
& \dfrac{\dfrac{KA}{x}\left( 200-0 \right)}{\dfrac{KA}{\left( 3.1-x \right)}\left( 200-100 \right)}=\dfrac{m\times {{s}_{ice}}}{m\times {{s}_{water}}} \\
& \dfrac{620-200x}{100x}=\dfrac{{{s}_{ice}}}{{{s}_{water}}}......(3) \\
\end{align}\]
Since it is mentioned that Latent heat of fusion of ice and vaporisation of water are 80 cal/gm and 540 cal/gm respectively
So, we can write equation (3) as:
$\begin{align}
& \dfrac{620-200x}{100 x}=\dfrac{80}{540} \\
& \dfrac{31-10x}{5x}=\dfrac{4}{27} \\
& 837-270x=20x \\
& 290x=837 \\
& x=2.88m \\
\end{align}$
So, the length of point from steam chamber is
$\begin{align}
& \left( 3.1-x \right)=3.1-2.88 \\
& =0.22m \\
& =22cm
\end{align}$
So, the correct answer is “Option B”.
Note:
Latent heat or specific heat is defined as the heat or energy that is absorbed or released during a phase change of a substance. It could either be from a gas to a liquid or liquid to solid and vice versa. Latent heat is related to a heat property called enthalpy.
Complete step by step answer:
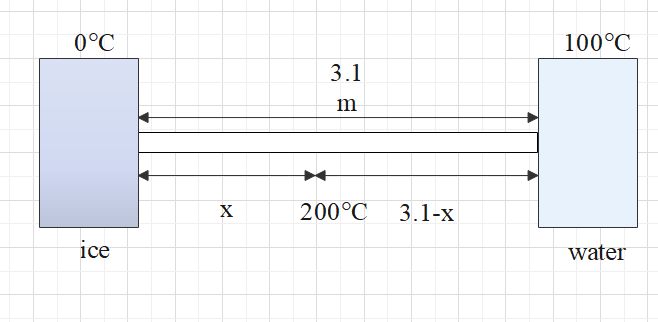
The heat rate by using the formula $Q=\dfrac{KA}{l}\Delta T=m\times s$
For ice:
\[{{Q}_{1}}=\dfrac{KA}{x}\left( 200-0 \right)=m\times {{s}_{ice}}......(1)\]
For water:
${{Q}_{2}}=\dfrac{KA}{\left( 3.1-x \right)}\left( 200-100 \right)=m\times {{s}_{water}}......(2)$
Divide equation (1) by (2), we get:
\[\begin{align}
& \dfrac{\dfrac{KA}{x}\left( 200-0 \right)}{\dfrac{KA}{\left( 3.1-x \right)}\left( 200-100 \right)}=\dfrac{m\times {{s}_{ice}}}{m\times {{s}_{water}}} \\
& \dfrac{620-200x}{100x}=\dfrac{{{s}_{ice}}}{{{s}_{water}}}......(3) \\
\end{align}\]
Since it is mentioned that Latent heat of fusion of ice and vaporisation of water are 80 cal/gm and 540 cal/gm respectively
So, we can write equation (3) as:
$\begin{align}
& \dfrac{620-200x}{100 x}=\dfrac{80}{540} \\
& \dfrac{31-10x}{5x}=\dfrac{4}{27} \\
& 837-270x=20x \\
& 290x=837 \\
& x=2.88m \\
\end{align}$
So, the length of point from steam chamber is
$\begin{align}
& \left( 3.1-x \right)=3.1-2.88 \\
& =0.22m \\
& =22cm
\end{align}$
So, the correct answer is “Option B”.
Note:
Latent heat or specific heat is defined as the heat or energy that is absorbed or released during a phase change of a substance. It could either be from a gas to a liquid or liquid to solid and vice versa. Latent heat is related to a heat property called enthalpy.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




