Nitration of phenol is an example of:
(a)- Nucleophilic addition
(b)- Nucleophilic substitution
(c)- Electrophilic substitution
(d)- Electrophilic addition
Answer
562.4k+ views
Hint: When nitric acid reacts with phenol either nitrophenol or picric acid is formed based on the reactants taken. Nitro molecule ($N{{O}_{2}}$) is an electrophile. The nitro group is attached to the phenol by removing the hydrogen atom.
Complete step by step answer:
Phenols undergo an Electrophilic substitution reaction. An example of an Electrophilic substitution reaction is the nitration of phenol.
There are 2 processes of nitration of phenol:
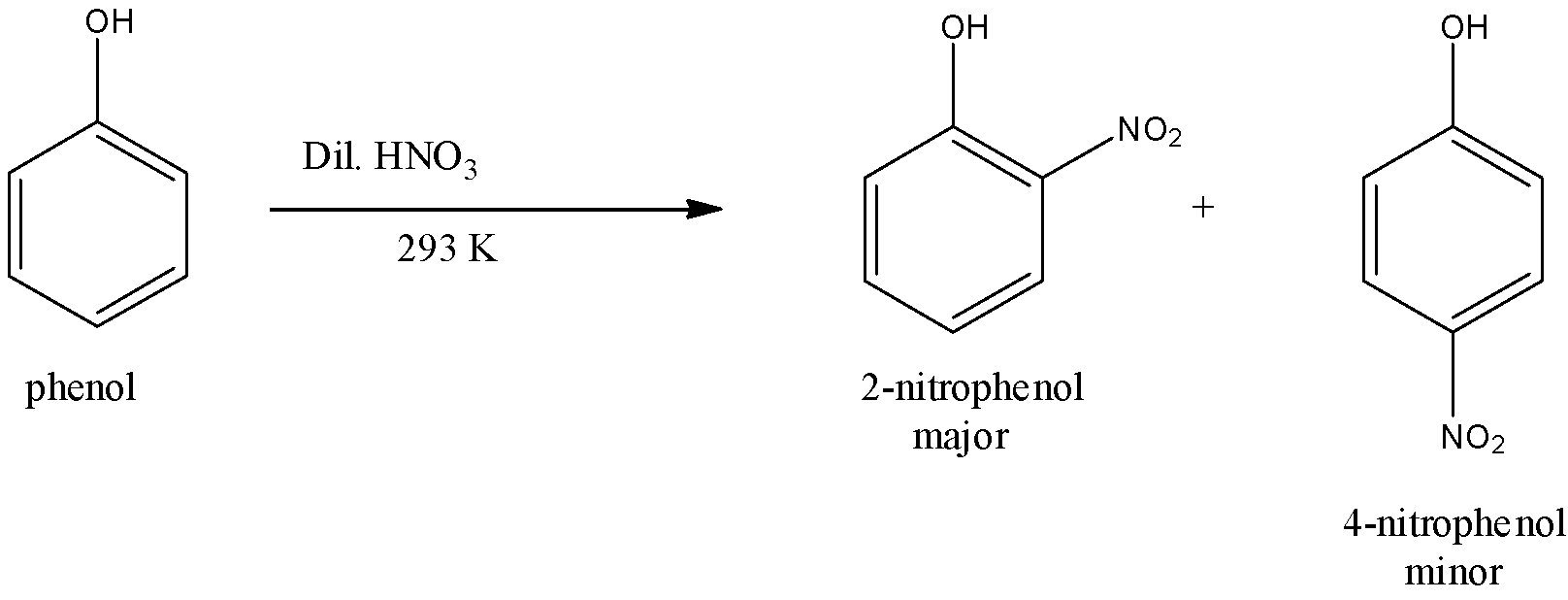
(i)- With dilute nitric acid: When dilute nitric acid at 293 K is used, phenols give mononitrophenols i.e., a mixture of 2-nitrophenol and 4-nitrophenol. Nitro molecule ($N{{O}_{2}}$) is an electrophile. The nitro group is attached to the phenol by removing the hydrogen atom. However, 2-nitrophenol predominates over 4-nitrophenol probably due to the stabilization of the transition state leading to the formation of 2-nitrophenol due to the intramolecular H-bonding. The reaction is given below:
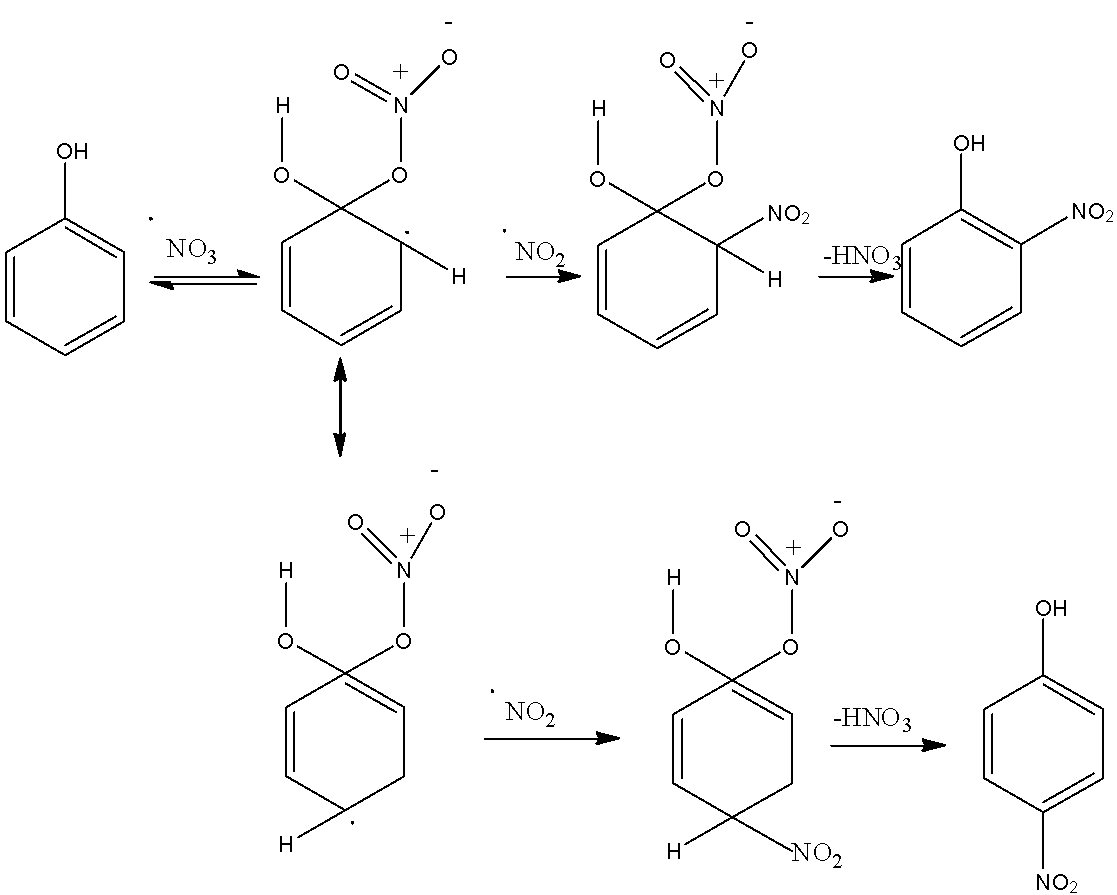
The mechanism of nitration is given below:
The intramolecular hydrogen bonding of 2-nitrophenol is given below:
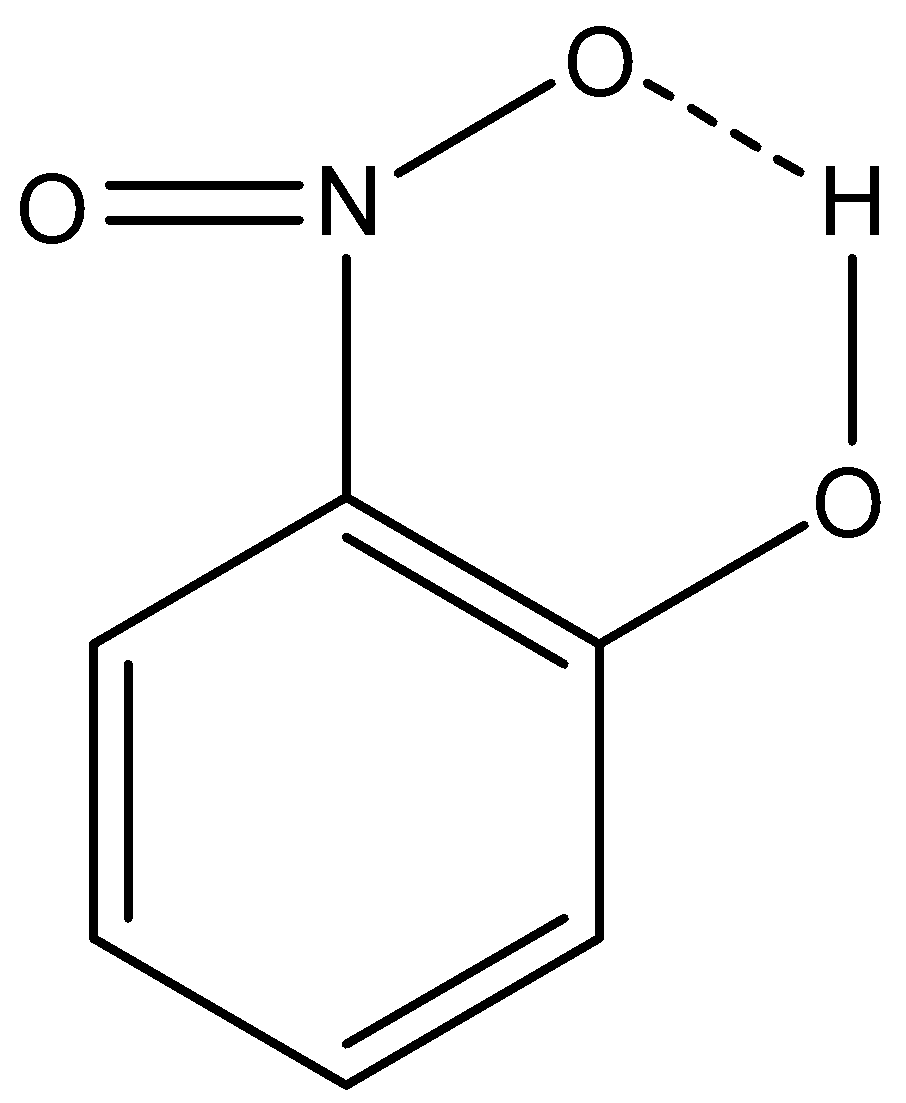
This intramolecular hydrogen bonding is not possible in 4-nitrophenol.
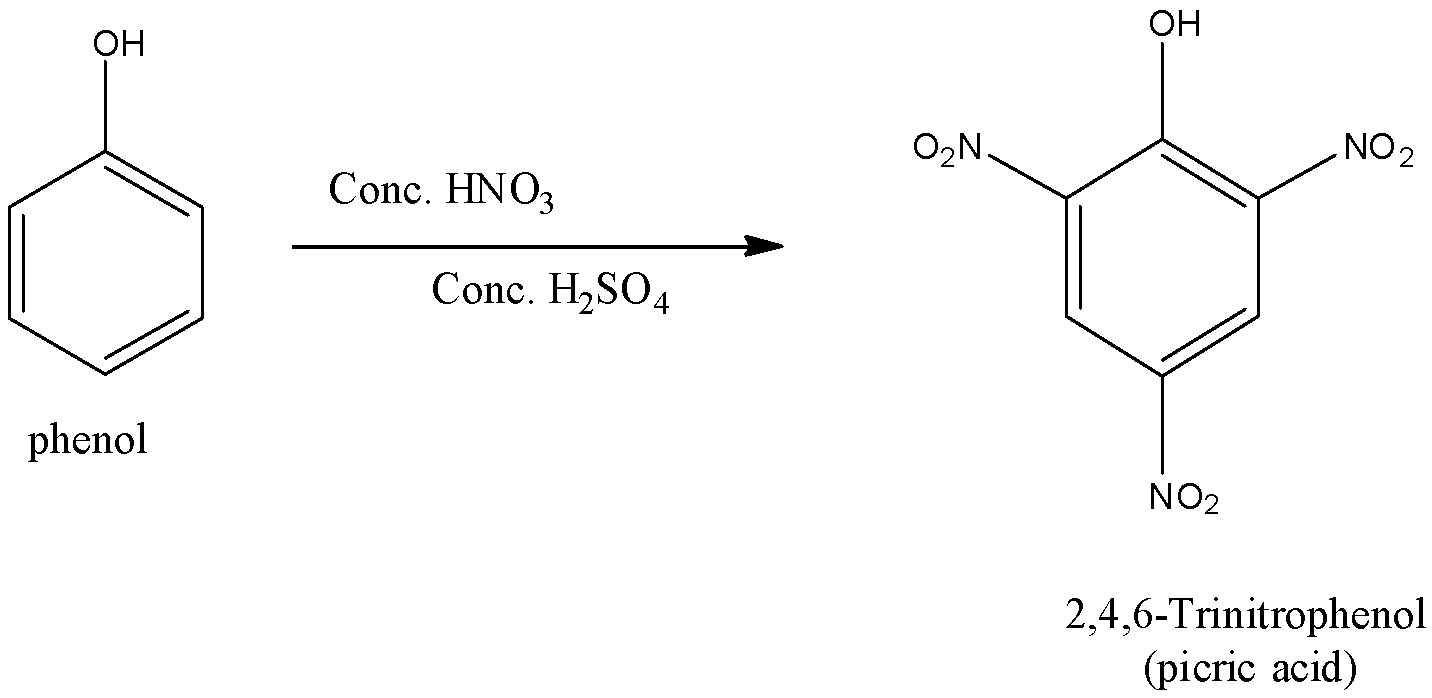
(ii)- With concentrated nitric acid: When the phenol is reacted with concentrated nitric acid in the presence of concentrated sulfuric acid gives a trisubstituted phenol called 2,4,6-Trinitrphenol or it is commonly called picric acid. The yield is poor since most of the phenol is oxidized by concentrated nitric acid. The reaction is given below:
So, the correct answer is an option (c)- Electrophilic substitution.
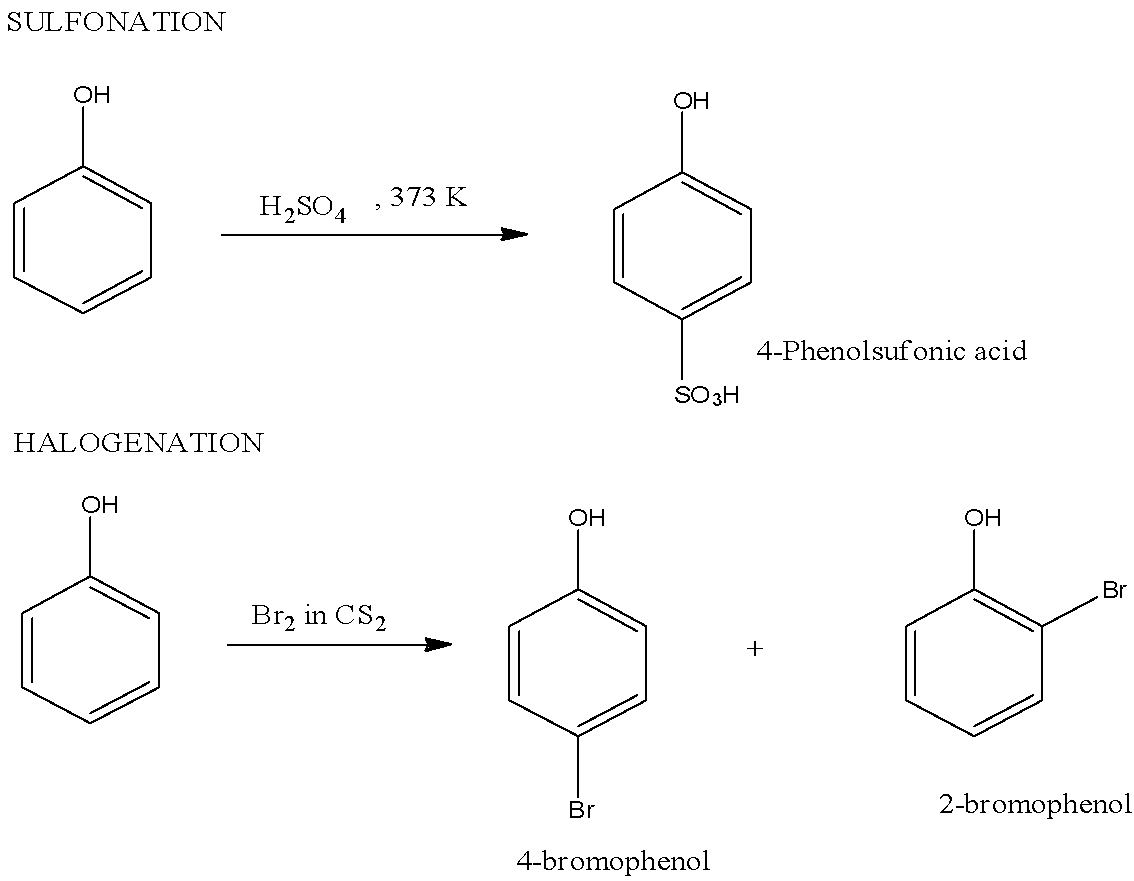
Note: Other examples of Electrophilic substitution reactions of phenol are halogenations in which halophenols are formed, sulfonation in which 4-Phenolsulfonic acid is formed, etc. The reaction of halogenations and sulfonation is given below:
Complete step by step answer:
Phenols undergo an Electrophilic substitution reaction. An example of an Electrophilic substitution reaction is the nitration of phenol.
There are 2 processes of nitration of phenol:
(i)- With dilute nitric acid: When dilute nitric acid at 293 K is used, phenols give mononitrophenols i.e., a mixture of 2-nitrophenol and 4-nitrophenol. Nitro molecule ($N{{O}_{2}}$) is an electrophile. The nitro group is attached to the phenol by removing the hydrogen atom. However, 2-nitrophenol predominates over 4-nitrophenol probably due to the stabilization of the transition state leading to the formation of 2-nitrophenol due to the intramolecular H-bonding. The reaction is given below:

The mechanism of nitration is given below:

The intramolecular hydrogen bonding of 2-nitrophenol is given below:

This intramolecular hydrogen bonding is not possible in 4-nitrophenol.
(ii)- With concentrated nitric acid: When the phenol is reacted with concentrated nitric acid in the presence of concentrated sulfuric acid gives a trisubstituted phenol called 2,4,6-Trinitrphenol or it is commonly called picric acid. The yield is poor since most of the phenol is oxidized by concentrated nitric acid. The reaction is given below:

So, the correct answer is an option (c)- Electrophilic substitution.
Note: Other examples of Electrophilic substitution reactions of phenol are halogenations in which halophenols are formed, sulfonation in which 4-Phenolsulfonic acid is formed, etc. The reaction of halogenations and sulfonation is given below:

Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




