Name the first organic compound which was obtained from an inorganic compound with formulae.
Answer
640.8k+ views
Hint: It was earlier believed that it is impossible to create organic compounds from inorganic compounds and that organic compounds like cells only arise from preexisting organic substances. It was only in the year 1828 this misconception was broken by a German chemist.
Complete step by step solution:
In 1828, the German chemist Friedrich Wöhler obtained urea artificially by treating silver cyanate with ammonium chloride.
\[AgNCO+N{{H}_{4}}Cl\,\,\to \,{{\left( N{{H}_{2}} \right)}_{2}}CO\,+\,AgCl\]
where \[{{\left( N{{H}_{2}} \right)}_{2}}CO\] is the formula of Urea.
This was the first time an organic compound was artificially synthesized from inorganic starting materials, without the involvement of living organisms. The results of this experiment implicitly discredited vitalism — the theory that the chemicals of living organisms are fundamentally different from those of inanimate matter.
It was earlier believed that Organic compounds can only be produced from other organic compounds.
Structure of Urea:
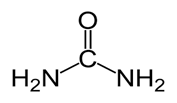
Thus Urea was the first organic compound to be synthesized by inorganic compounds.
Additional Information:
Urea is also known as carbamide since there is a carbonyl group attached to two amide groups. Urea is a highly soluble organic compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids.
Note: Note that isolation of organic compounds can be traced extremely far back in history. The ancient Egyptians isolated indigo as a colouring agent. However, they didn’t synthesize it and most attempts pre-19th century failed. Nobody questioned the possibility of isolating pure organic compounds from living matter, it was merely questioned whether they could be synthesized without involvement of organisms.
Wohler’s discovery was that significant, because he used compounds that were (and are) unanimously declared inorganic (silver cyanate and ammonium chloride) and arrived directly at a compound that had previously been isolated from living material namely urea. Sometimes in Question they also say that urea is also prepared from ammonium cyanate.
Complete step by step solution:
In 1828, the German chemist Friedrich Wöhler obtained urea artificially by treating silver cyanate with ammonium chloride.
\[AgNCO+N{{H}_{4}}Cl\,\,\to \,{{\left( N{{H}_{2}} \right)}_{2}}CO\,+\,AgCl\]
where \[{{\left( N{{H}_{2}} \right)}_{2}}CO\] is the formula of Urea.
This was the first time an organic compound was artificially synthesized from inorganic starting materials, without the involvement of living organisms. The results of this experiment implicitly discredited vitalism — the theory that the chemicals of living organisms are fundamentally different from those of inanimate matter.
It was earlier believed that Organic compounds can only be produced from other organic compounds.
Structure of Urea:

Thus Urea was the first organic compound to be synthesized by inorganic compounds.
Additional Information:
Urea is also known as carbamide since there is a carbonyl group attached to two amide groups. Urea is a highly soluble organic compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids.
Note: Note that isolation of organic compounds can be traced extremely far back in history. The ancient Egyptians isolated indigo as a colouring agent. However, they didn’t synthesize it and most attempts pre-19th century failed. Nobody questioned the possibility of isolating pure organic compounds from living matter, it was merely questioned whether they could be synthesized without involvement of organisms.
Wohler’s discovery was that significant, because he used compounds that were (and are) unanimously declared inorganic (silver cyanate and ammonium chloride) and arrived directly at a compound that had previously been isolated from living material namely urea. Sometimes in Question they also say that urea is also prepared from ammonium cyanate.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




