Molecule having a non zero dipole moment?
a) $ S{F_4} $
b) $ Si{F_4} $
c) $ Xe{F_4} $
d) $ B{F_3}^{} $
Answer
545.1k+ views
Hint: The dipole moment is the product of negative or positive charge and the distance between the centers of positive and negative charges. The dipole moment is directly towards the more electronegative atom in the compound. The opposite direction of the dipole moment in the molecule cancels each other.
Complete answer:
Let us see all the options.
The structure of $ Si{F_4} $ is :
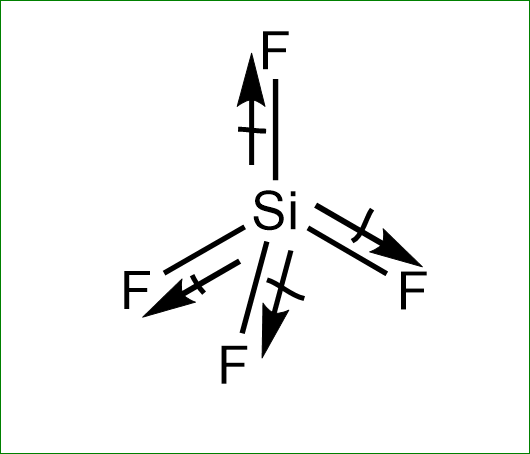
The fluorine is more electronegative than the silicon atom. hence, the dipole moment will direct towards the fluorine atom. Since the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other three fluorine. Hence it has zero dipole moment.
ii) $ Xe{F_4} $
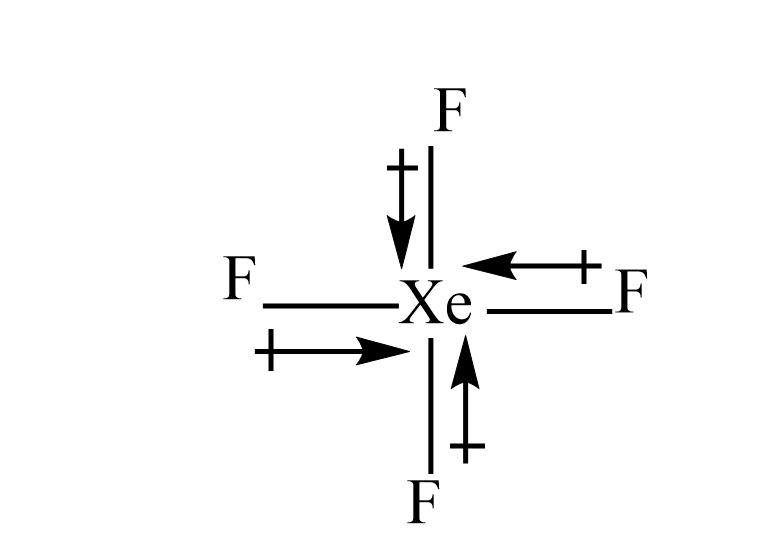
The fluorine is more electronegative than the xenon atom. hence, the dipole moment will direct towards the fluorine atom. Since, the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other three fluorine. Hence even this will have zero dipole moment.
iii) $ S{F_4} $
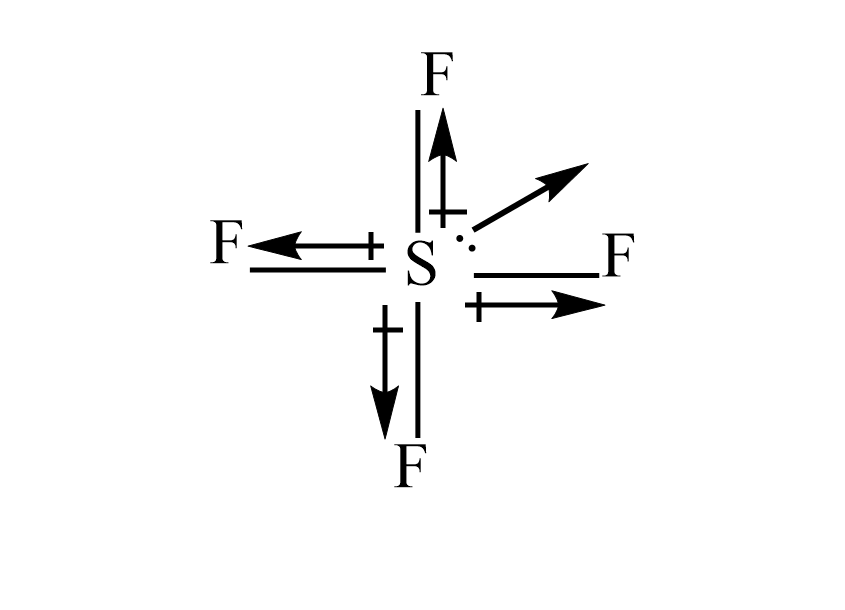
The fluorine is more electronegative than the sulphur atom. so, the dipole moment will direct towards the fluorine atom. Since the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other fluorine, and the lone pair does not get cancelled out. Hence it has a non zero dipole moment.
iv) $ B{F_3}^{} $
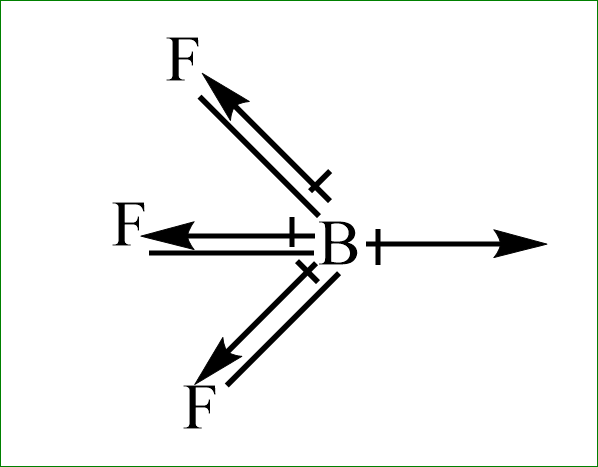
Even here, the fluorine is more electronegative than the boron atom. the dipole moment will direct towards the fluorine atom. Since, the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other two fluorine. Hence this will have zero dipole moment.
Note:
The dipole moment is the product of the positive or negative charge and the distance between centers of the positive and negative charges. It is denoted by the dipole moment directed towards the more electronegative atom.
Complete answer:
Let us see all the options.
The structure of $ Si{F_4} $ is :

The fluorine is more electronegative than the silicon atom. hence, the dipole moment will direct towards the fluorine atom. Since the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other three fluorine. Hence it has zero dipole moment.
ii) $ Xe{F_4} $

The fluorine is more electronegative than the xenon atom. hence, the dipole moment will direct towards the fluorine atom. Since, the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other three fluorine. Hence even this will have zero dipole moment.
iii) $ S{F_4} $

The fluorine is more electronegative than the sulphur atom. so, the dipole moment will direct towards the fluorine atom. Since the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other fluorine, and the lone pair does not get cancelled out. Hence it has a non zero dipole moment.
iv) $ B{F_3}^{} $

Even here, the fluorine is more electronegative than the boron atom. the dipole moment will direct towards the fluorine atom. Since, the dipole moment of one fluorine atom is cancelled by resultant dipole moments of the other two fluorine. Hence this will have zero dipole moment.
Note:
The dipole moment is the product of the positive or negative charge and the distance between centers of the positive and negative charges. It is denoted by the dipole moment directed towards the more electronegative atom.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




