How to make these molecules from cyclopentene?
Starting from cyclopentene how would you prepare this molecules:
Answer
545.7k+ views
Hint: We must have to know that the cyclopentene is an organic compound having cyclic structure which is formed by five carbon atoms in a ring having a double bond. Many conversions can be carried out using these molecules. We can look for the easiest method that can be single step so as to derive the following compounds or conversions.
Complete answer:
a).Chlorocyclopentane
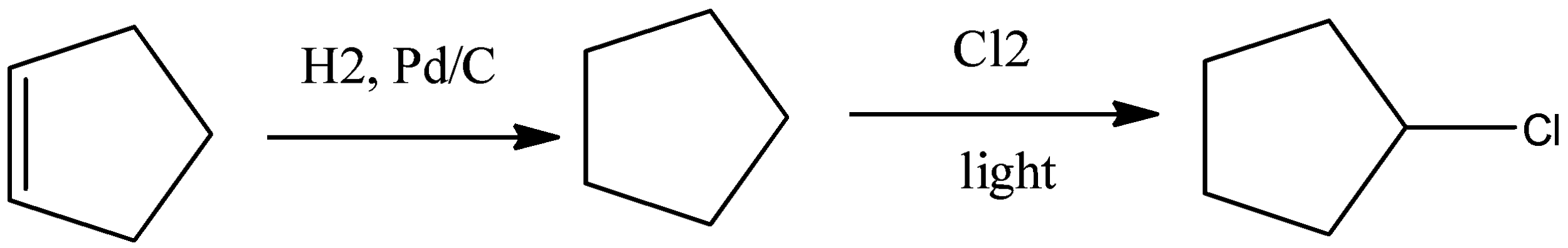
Cyclopentene was first reduced to cyclopentane using palladium catalyst and carbon and hydrogen gas. Then in the presence of light and chlorine molecules it gets converted to chlorocyclopentane.
b).3-bromocyclopentane
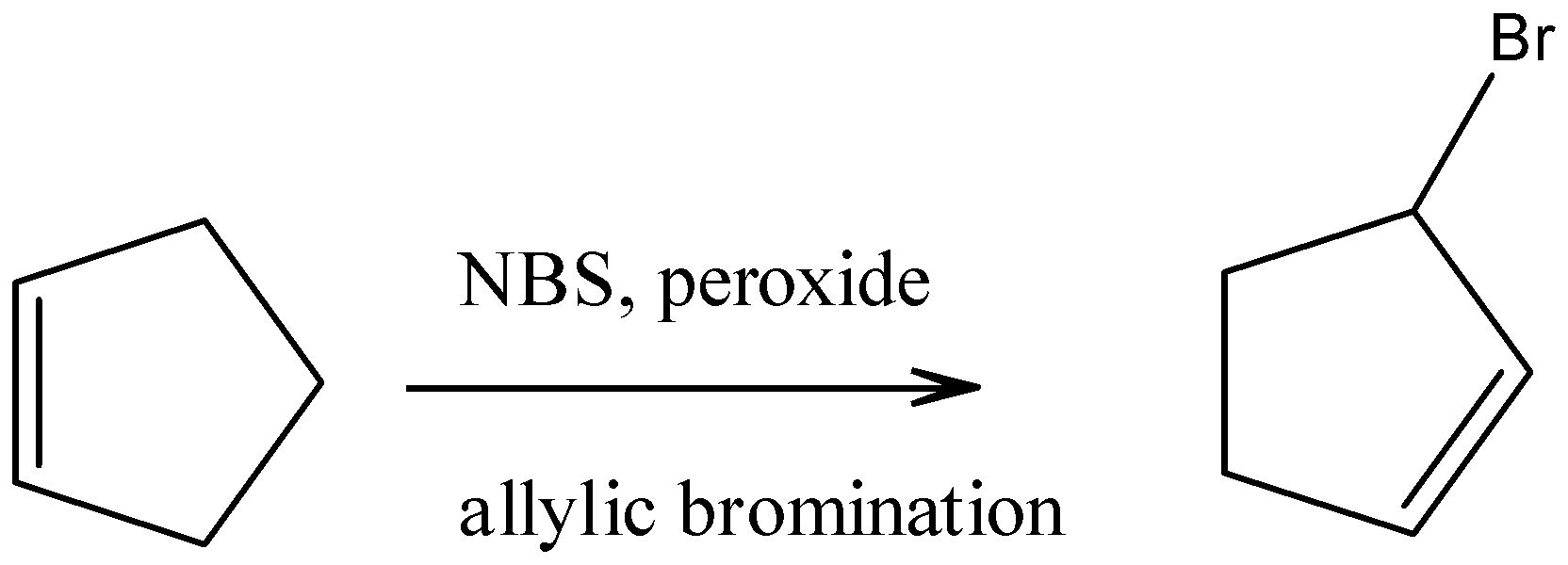
On allylic bromination of cyclopentene with n bromosuccinimide and peroxide we get 3-bromocyclopentene.
c).Cyclopentanol
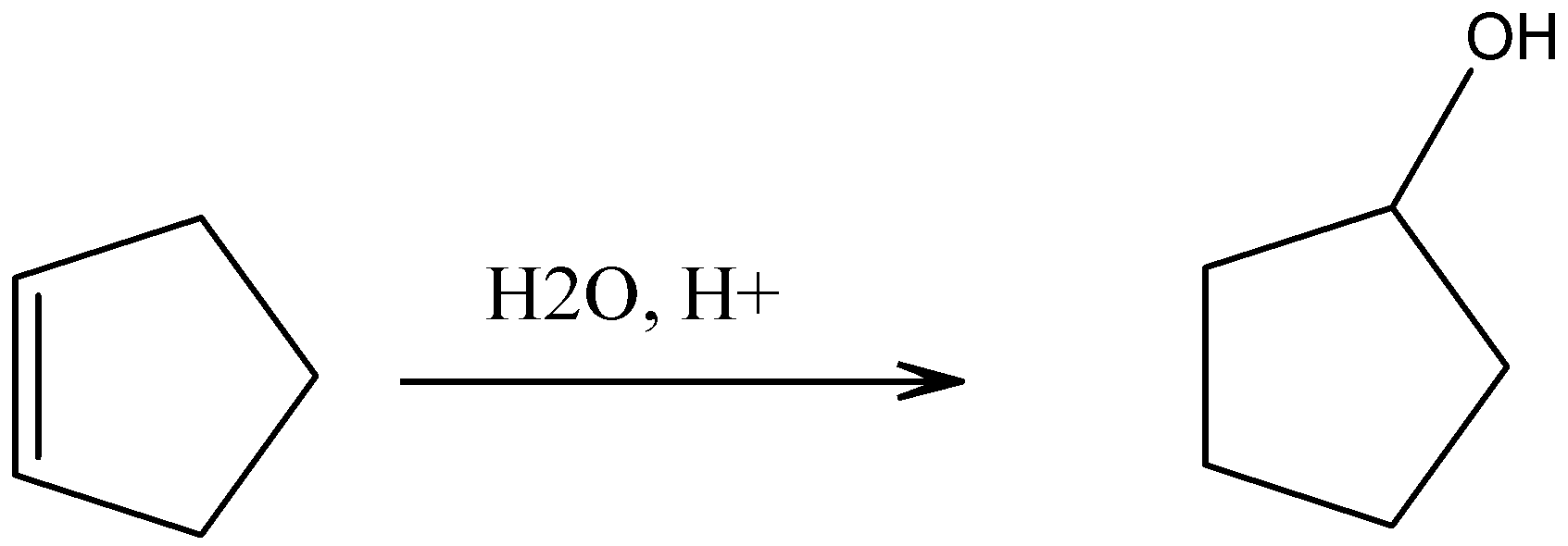
On hydrolysis of cyclopentene, we can get cyclopentanol, which is an alcohol.
d).Cyclopentylcyclopentane
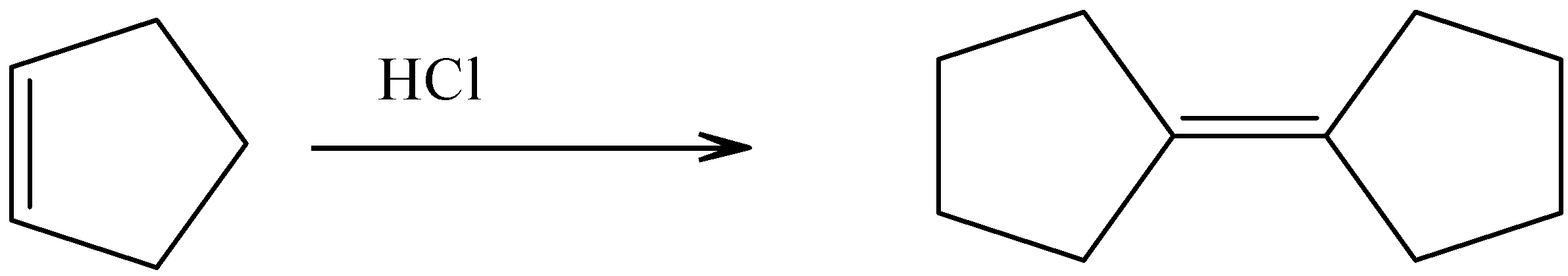
In presence of a strong acid like hydrochloric acid we can convert cyclopentene to cyclopentylcyclopentane structure.
Note:
Cyclopentene is a chemical compound with the formula (C{H_3})(C{H_2}). It is a colorless liquid with a petrol-like odor. It is one of the cycloalkenes. Cyclopentene is produced industrially in large amounts by steam cracking of naphtha. It has few applications, and thus is mainly used as a component of gasoline. We must know that cyclopentene is produced industrially in large amounts. It is used as a monomer for synthesis of plastics, and in a number of chemical syntheses. It is a colorless liquid with a petrol-like odor.
Complete answer:
a).Chlorocyclopentane

Cyclopentene was first reduced to cyclopentane using palladium catalyst and carbon and hydrogen gas. Then in the presence of light and chlorine molecules it gets converted to chlorocyclopentane.
b).3-bromocyclopentane

On allylic bromination of cyclopentene with n bromosuccinimide and peroxide we get 3-bromocyclopentene.
c).Cyclopentanol

On hydrolysis of cyclopentene, we can get cyclopentanol, which is an alcohol.
d).Cyclopentylcyclopentane

In presence of a strong acid like hydrochloric acid we can convert cyclopentene to cyclopentylcyclopentane structure.
Note:
Cyclopentene is a chemical compound with the formula (C{H_3})(C{H_2}). It is a colorless liquid with a petrol-like odor. It is one of the cycloalkenes. Cyclopentene is produced industrially in large amounts by steam cracking of naphtha. It has few applications, and thus is mainly used as a component of gasoline. We must know that cyclopentene is produced industrially in large amounts. It is used as a monomer for synthesis of plastics, and in a number of chemical syntheses. It is a colorless liquid with a petrol-like odor.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




