Lateral overlap of p-orbitals leads to the formation of ______.
A) ${{\pi }}$-bond
B) Metallic bond
C) Sigma bond
D) Ionic bond
Answer
593.7k+ views
Hint:We know that the bonds are formed by the overlapping of orbitals. The extent of orbital overlapping depends on the size of the two participating atoms, and the valence electrons. The concentration of orbitals on the adjacent atoms in the same region is known as orbital overlapping.
Complete answer :
We know that the bonds are formed by the overlapping of orbitals. The extent of orbital overlapping depends on the size of the two participating atoms, and the valence electrons. The concentration of orbitals on the adjacent atoms in the same region is known as orbital overlapping.
Orbital overlapping leads to the formation of two types of bonds:
i) Sigma bond
ii) pi-bond
The covalent bonds formed by the axial overlap of s-orbitals are known as $\sigma $-bonds (sigma bond). The covalent bonds formed by the lateral overlap of p-orbitals or the half-filled atomic orbitals are known as ${{\pi }}$-bonds (pi bonds).
The formation of ${{\pi }}$-bond occurs as follows:
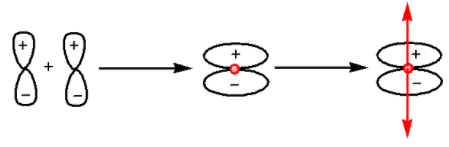
From the diagram we can see that ${{\pi }}$-bond is unsymmetrical.Thus, lateral overlap of p-orbitals leads to the formation of ${{\pi }}$-bond.
Therefore, the correct answer is option (A) ${{\pi }}$-bond.
Note:The extent of overlapping in ${{\pi }}$-bond is very less and thus, ${{\pi }}$-bond is weak in nature. In ${{\pi }}$-bond, the electron density is concentrated in the region which is perpendicular to the axis of the bond. A double covalent bond exists when one ${{\pi }}$-bond and one $\sigma $-bond is formed. A triple covalent bond exists when two ${{\pi }}$-bond and one $\sigma $-bond is formed.
Complete answer :
We know that the bonds are formed by the overlapping of orbitals. The extent of orbital overlapping depends on the size of the two participating atoms, and the valence electrons. The concentration of orbitals on the adjacent atoms in the same region is known as orbital overlapping.
Orbital overlapping leads to the formation of two types of bonds:
i) Sigma bond
ii) pi-bond
The covalent bonds formed by the axial overlap of s-orbitals are known as $\sigma $-bonds (sigma bond). The covalent bonds formed by the lateral overlap of p-orbitals or the half-filled atomic orbitals are known as ${{\pi }}$-bonds (pi bonds).
The formation of ${{\pi }}$-bond occurs as follows:

From the diagram we can see that ${{\pi }}$-bond is unsymmetrical.Thus, lateral overlap of p-orbitals leads to the formation of ${{\pi }}$-bond.
Therefore, the correct answer is option (A) ${{\pi }}$-bond.
Note:The extent of overlapping in ${{\pi }}$-bond is very less and thus, ${{\pi }}$-bond is weak in nature. In ${{\pi }}$-bond, the electron density is concentrated in the region which is perpendicular to the axis of the bond. A double covalent bond exists when one ${{\pi }}$-bond and one $\sigma $-bond is formed. A triple covalent bond exists when two ${{\pi }}$-bond and one $\sigma $-bond is formed.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

The highest temperature in Karnataka is recorded in class 10 social science CBSE




