What kind of reaction breaks down polymers into monomers?
Answer
556.2k+ views
Hint: The monomers like amino acids combined through a peptide bond to form a polypeptide chain and later the polypeptide chains combined and forms a protein structure. At the same time the polypeptide chain undergoes breakdown and forms monomers amino acids as the products.
Complete answer:
- In the question it is asked what type of reaction breaks down the polymers into monomers.
- Almost the formation of all the polymers occurs through the release of the water molecules as the byproduct.
- By using the same byproduct we can break the formed bond and the chemical reaction is called hydrolysis.
- For example take a dimer of carbohydrate and there is a glycosidic bond in between the glucose and galactose.
- The cleavage or the hydrolysis of lactose is going to give two monomers called glucose and galactose.
- We can see the hydrolysis of the lactose means conversion of a dimer into two monomers as shown in below chemical reaction hydrolysis of lactose.
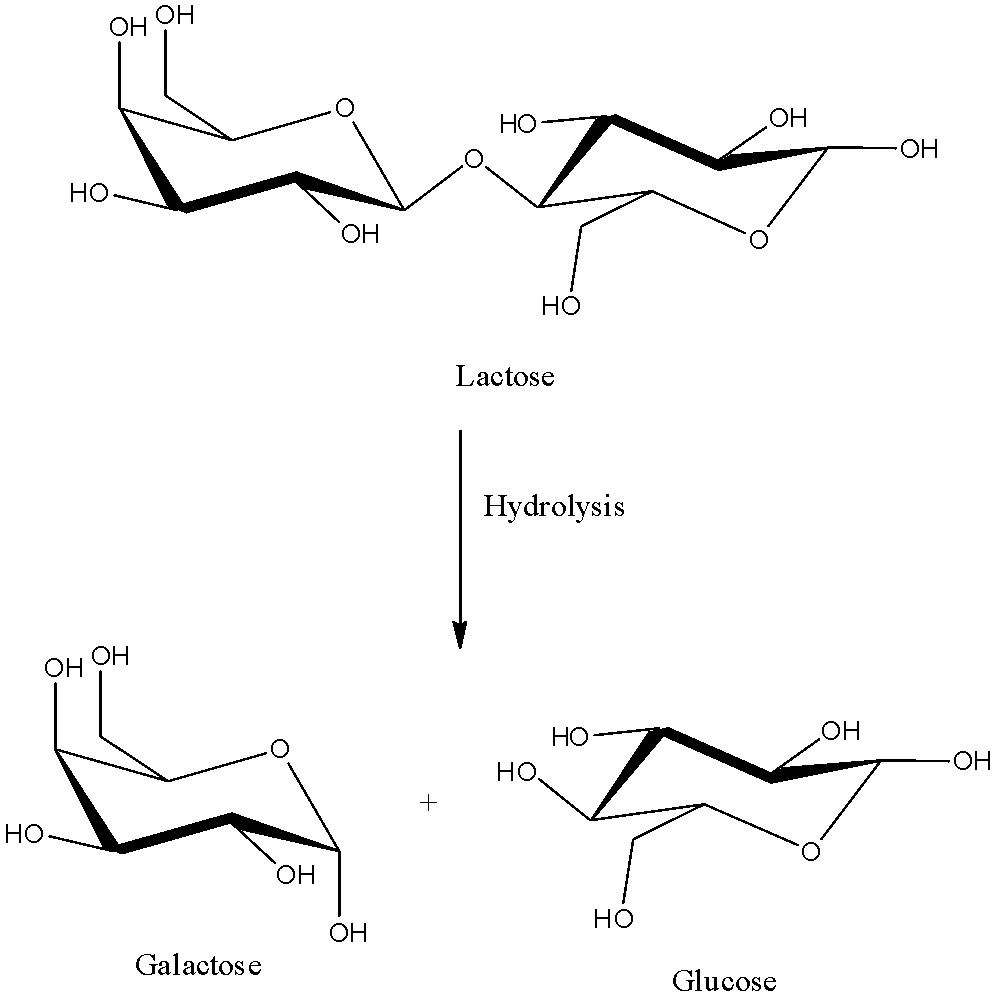
- Hydrolysis is going to break the bond between the monomers in the polymeric chain.
- The process of breakdown of the polymers into monomers is called hydrolysis or the cleavage of the bond between the monomers in a polymeric chain.
Note:
The opposite to hydrolysis is dehydration which is going to remove the water and forms a bond between the monomers to form the process of the preparation of the polymer and the process is called polymerization.
Complete answer:
- In the question it is asked what type of reaction breaks down the polymers into monomers.
- Almost the formation of all the polymers occurs through the release of the water molecules as the byproduct.
- By using the same byproduct we can break the formed bond and the chemical reaction is called hydrolysis.
- For example take a dimer of carbohydrate and there is a glycosidic bond in between the glucose and galactose.
- The cleavage or the hydrolysis of lactose is going to give two monomers called glucose and galactose.
- We can see the hydrolysis of the lactose means conversion of a dimer into two monomers as shown in below chemical reaction hydrolysis of lactose.

- Hydrolysis is going to break the bond between the monomers in the polymeric chain.
- The process of breakdown of the polymers into monomers is called hydrolysis or the cleavage of the bond between the monomers in a polymeric chain.
Note:
The opposite to hydrolysis is dehydration which is going to remove the water and forms a bond between the monomers to form the process of the preparation of the polymer and the process is called polymerization.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




