
What is the IUPAC name of \[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\]?
A. Tetracyanonickelate\[\left( {II} \right)\]
B. Tetracyanonickel\[\left( {II} \right)\]
C. Tetracyanonickelate\[\left( I \right)\]
D. None of these
Answer
513.6k+ views
Hint: The IUPAC nomenclature is a chemical nomenclature which is used for the naming of organic compounds by following some rules and regulations. And it is proposed by the International Union of Pure and Applied Chemistry. For solving an IUPAC name, first we have to identify the longest carbon chain present in the molecule and look if any functional is there or any branched chain is there or not. After that, conclude the name into a single word.
Complete answer:
The IUPAC name of \[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\] is tetracyanonickelate\[\left( {II} \right)\]. Here, the central metal atom is nickel. The nickel atom is linked with four $ - CN$ (cyano) groups, so it should contain ‘tetracyano’. And the charge of the complex is \[ - 2\]. Thus, it is required to write in a bracket, \[\left( {II} \right)\]. The central atom should contain a suffix which is ‘ate’. Therefore, the IUPAC name of the complex is tetracyanonickelate\[\left( {II} \right)\]. Let’s see the chemical structure,
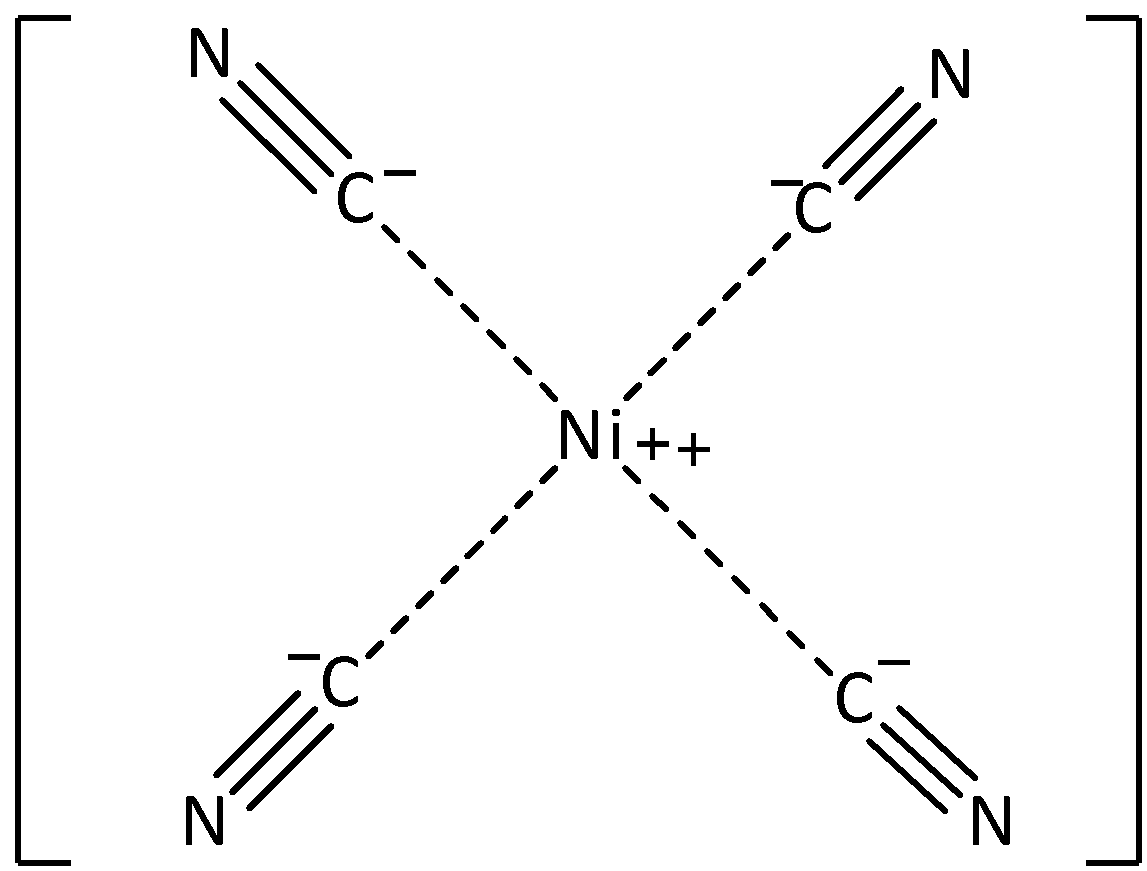
Hence, option (A) is correct.
The name should contain ‘ate’ with their central metal atom. Hence, option (B) is incorrect.
The charge of the complex is \[ - 2\]. Hence, option (C) is incorrect.
The IUPAC name of the complex is tetracyanonickelate\[\left( {II} \right)\]. Hence, option (D) is incorrect.
So, the correct answer is “Option A”.
Note:
The IUPAC nomenclature is used for the naming of organic compounds. For that, first we have to identify the longest continuous carbon chain present in the parent compound and find out the group linked to this longest chain. The name is different for each functional group. For example, if the acid group is present in the compound, the suffix should be ‘-oate’. Like that, we are naming an organic compound.
Complete answer:
The IUPAC name of \[{\left[ {Ni{{\left( {CN} \right)}_4}} \right]^{2 - }}\] is tetracyanonickelate\[\left( {II} \right)\]. Here, the central metal atom is nickel. The nickel atom is linked with four $ - CN$ (cyano) groups, so it should contain ‘tetracyano’. And the charge of the complex is \[ - 2\]. Thus, it is required to write in a bracket, \[\left( {II} \right)\]. The central atom should contain a suffix which is ‘ate’. Therefore, the IUPAC name of the complex is tetracyanonickelate\[\left( {II} \right)\]. Let’s see the chemical structure,

Hence, option (A) is correct.
The name should contain ‘ate’ with their central metal atom. Hence, option (B) is incorrect.
The charge of the complex is \[ - 2\]. Hence, option (C) is incorrect.
The IUPAC name of the complex is tetracyanonickelate\[\left( {II} \right)\]. Hence, option (D) is incorrect.
So, the correct answer is “Option A”.
Note:
The IUPAC nomenclature is used for the naming of organic compounds. For that, first we have to identify the longest continuous carbon chain present in the parent compound and find out the group linked to this longest chain. The name is different for each functional group. For example, if the acid group is present in the compound, the suffix should be ‘-oate’. Like that, we are naming an organic compound.
Recently Updated Pages
Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Draw a diagram of nephron and explain its structur class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

Chemical formula of Bleaching powder is A Ca2OCl2 B class 11 chemistry CBSE

Name the part of the brain responsible for the precision class 11 biology CBSE

The growth of tendril in pea plants is due to AEffect class 11 biology CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE




