IUPAC name of
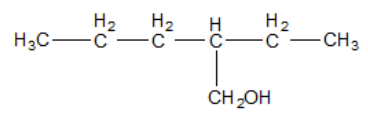
A. 3-propyl butan-1-ol
B. 2-ethylpentan-1-ol
C. 3-methyl hydroxy hexane
D. 2-ethyl-2-propyl ethanol

Answer
564.6k+ views
Hint: To answer this question, you should recall the concept of IUPAC nomenclature of IUPAC compounds. The IUPAC system rose to the necessity for a systematic approach due to the sheer quantity of discoveries of organic compounds which made the vernacular nomenclature of organic compounds highly inconvenient.
Complete step-by-step answer:
According to the Guidelines set by IUPAC, the nomenclature of compounds must follow these steps:
1.The Longest Chain Rule
2.The Lowest Set of Locants: Numbering of parent chains in which the lowest number is assigned to the carbon atom which carries the substituents.
3.Multiple instances of the same substituent
4.The naming of different substituents
5.Correct naming of different substituents in case they are present at the same positions
6.Naming Complex Substituents
7.The longest carbon chain is composed of 6 carbon chains and the substituent is methyl hydroxyl group.
Hence, the systematic name of the given compound is 3-methyl hydroxy hexane, i.e. option C.
Note: You should know about different types of isomerism and their naming. We know that isomers are defined as the molecules with the same molecular formula but possess a different arrangement of the atoms in space or different connectivity of atoms. The phenomenon in which the molecules in which the atoms that form the isomers are connected differently is known as structural isomerism. The phenomenon in which the connectivity of atoms is the same in isomers but a different spatial arrangement is a stereoisomerism. 2-Butene can exist as cis and trans isomers because of the double bond that leads to the restricted rotation. For example, in 2-Butene, this results in two isomers where the cis-isomer formed have the two methyl groups on the same side and the trans-isomer formed has the two methyl groups on opposite sides. 2-methyl propene and 2-methyl-2-butene contain a double bond but the groups attached to one of the C of the double bond are the same.
Complete step-by-step answer:
According to the Guidelines set by IUPAC, the nomenclature of compounds must follow these steps:
1.The Longest Chain Rule
2.The Lowest Set of Locants: Numbering of parent chains in which the lowest number is assigned to the carbon atom which carries the substituents.
3.Multiple instances of the same substituent
4.The naming of different substituents
5.Correct naming of different substituents in case they are present at the same positions
6.Naming Complex Substituents
7.The longest carbon chain is composed of 6 carbon chains and the substituent is methyl hydroxyl group.
Hence, the systematic name of the given compound is 3-methyl hydroxy hexane, i.e. option C.
Note: You should know about different types of isomerism and their naming. We know that isomers are defined as the molecules with the same molecular formula but possess a different arrangement of the atoms in space or different connectivity of atoms. The phenomenon in which the molecules in which the atoms that form the isomers are connected differently is known as structural isomerism. The phenomenon in which the connectivity of atoms is the same in isomers but a different spatial arrangement is a stereoisomerism. 2-Butene can exist as cis and trans isomers because of the double bond that leads to the restricted rotation. For example, in 2-Butene, this results in two isomers where the cis-isomer formed have the two methyl groups on the same side and the trans-isomer formed has the two methyl groups on opposite sides. 2-methyl propene and 2-methyl-2-butene contain a double bond but the groups attached to one of the C of the double bond are the same.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

What is 1s 2s 2p 3s 3p class 11 chemistry CBSE




