In which of the following pairs, the two species are iso-structural?
A. \[BrO_3^ - \] and \[Xe{O_3}\]
B. \[S{F_4}\] and \[Xe{F_4}\]
C. \[SO_3^{2 - }\] and \[NO_3^ - \]
D. \[B{F_3}\] and \[N{F_3}\]
Answer
577.2k+ views
Hint: Iso-structural is defined as those species which contain the same number of atoms arranged in the same structural form which same number of chemical bonds and lone pairs of electrons..
Complete step by step answer:
A. \[BrO_3^ - \] and \[Xe{O_3}\]
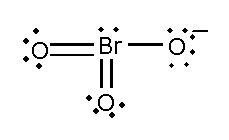
In this compound, the total number of atoms present in this structure is 4, lone pair present is 7, minus charge shows that it contains extra electrons.
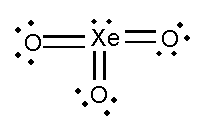
In this compound, the total number of atoms present in this structure is 4, lone pair present is 7.
Both the given structures are isostructural to each other.
B. \[S{F_4}\] and \[Xe{F_4}\]
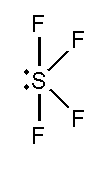
In this compound, the total number of atoms present in this structure is 4, lone pair present is 1.
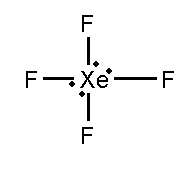
In this compound, the total number of atoms present in this structure is 4, lone pair present is 2.
Both the given compounds are not isostructural to each other.
C. \[SO_3^{2 - }\] and \[NO_3^ - \]
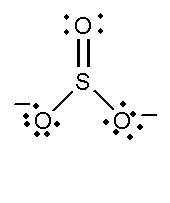
In this compound, the total number of atoms present in this structure is 4, lone pair present is 8. Two single bonds and one double bond is present. The total charge is -2.
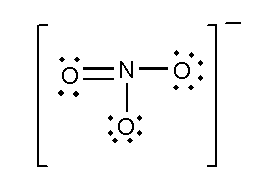
In this compound, the total number of atoms present in this structure is 4, lone pair present is 8. Two single bonds and one double bond is present. The total charge is -1.
Both the given compounds are not isostructural to each other.
D. D. \[B{F_3}\] and \[N{F_3}\]
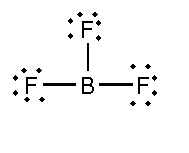
In this compound, the total number of atoms present is 4, the total number of lone pairs present is 9 and three single bonds are present.
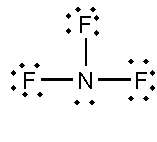
In this compound, the total number of atoms present is 4, the total number of lone pairs present is 10 and three single bonds are present.
Both the given compounds are not isostructural to each other.
So, the correct answer is Option A.
Note: The correct option can also be found by the formula of hybridization which is shown below.
\[ \Rightarrow \dfrac{1}{2}\left[ {no\;of\;valence\;{e^ - }on\;central\;atom + no\;of\;monovalent\;atom \pm ch\arg e} \right]\]
For \[BrO_3^ - \]
\[ \Rightarrow \dfrac{1}{2}\left[ {7 + 0 + 1} \right] = 4\;s{p^3}\]
For \[Xe{O_3}\]
\[ \Rightarrow \dfrac{1}{2}\left[ {8 + 0 \pm 0} \right] = 4\;s{p^3}\]
Complete step by step answer:
A. \[BrO_3^ - \] and \[Xe{O_3}\]

In this compound, the total number of atoms present in this structure is 4, lone pair present is 7, minus charge shows that it contains extra electrons.
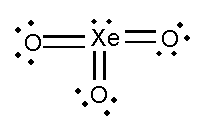
In this compound, the total number of atoms present in this structure is 4, lone pair present is 7.
Both the given structures are isostructural to each other.
B. \[S{F_4}\] and \[Xe{F_4}\]

In this compound, the total number of atoms present in this structure is 4, lone pair present is 1.
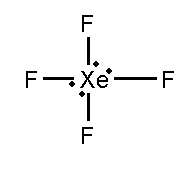
In this compound, the total number of atoms present in this structure is 4, lone pair present is 2.
Both the given compounds are not isostructural to each other.
C. \[SO_3^{2 - }\] and \[NO_3^ - \]

In this compound, the total number of atoms present in this structure is 4, lone pair present is 8. Two single bonds and one double bond is present. The total charge is -2.

In this compound, the total number of atoms present in this structure is 4, lone pair present is 8. Two single bonds and one double bond is present. The total charge is -1.
Both the given compounds are not isostructural to each other.
D. D. \[B{F_3}\] and \[N{F_3}\]

In this compound, the total number of atoms present is 4, the total number of lone pairs present is 9 and three single bonds are present.

In this compound, the total number of atoms present is 4, the total number of lone pairs present is 10 and three single bonds are present.
Both the given compounds are not isostructural to each other.
So, the correct answer is Option A.
Note: The correct option can also be found by the formula of hybridization which is shown below.
\[ \Rightarrow \dfrac{1}{2}\left[ {no\;of\;valence\;{e^ - }on\;central\;atom + no\;of\;monovalent\;atom \pm ch\arg e} \right]\]
For \[BrO_3^ - \]
\[ \Rightarrow \dfrac{1}{2}\left[ {7 + 0 + 1} \right] = 4\;s{p^3}\]
For \[Xe{O_3}\]
\[ \Rightarrow \dfrac{1}{2}\left[ {8 + 0 \pm 0} \right] = 4\;s{p^3}\]
Recently Updated Pages
The magnetic field in a plane electromagnetic wave class 11 physics CBSE

In a plane electromagnetic wave the electric field class 12 physics CBSE

A plane electromagnetic wave travels in vacuum along class 12 physics CBSE

Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

What are the examples of C3 and C4 plants class 11 biology CBSE

State and prove Bernoullis theorem class 11 physics CBSE

10 examples of friction in our daily life

A body is said to be in dynamic equilibrium if A When class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells




