In which of the following molecules all atoms are not coplanar?
- A.
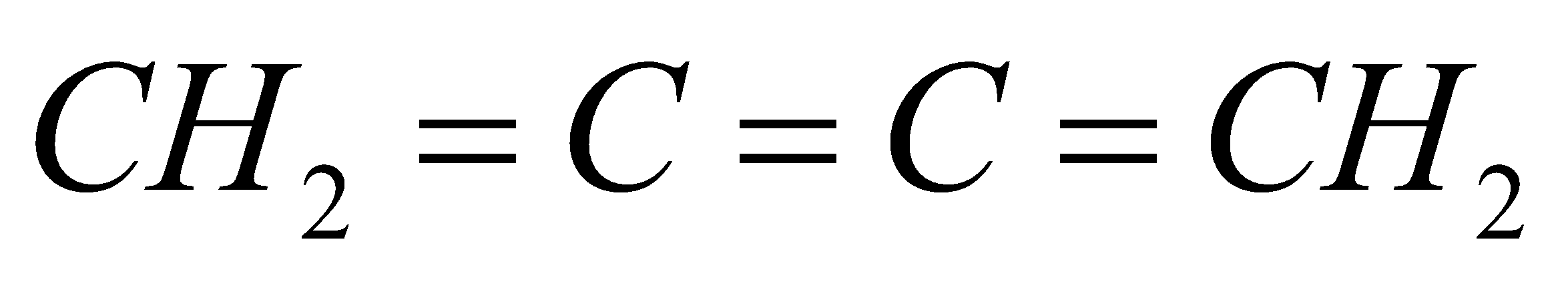
B.
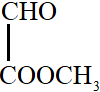
C.
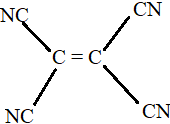
D.
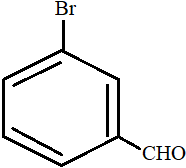
- A.
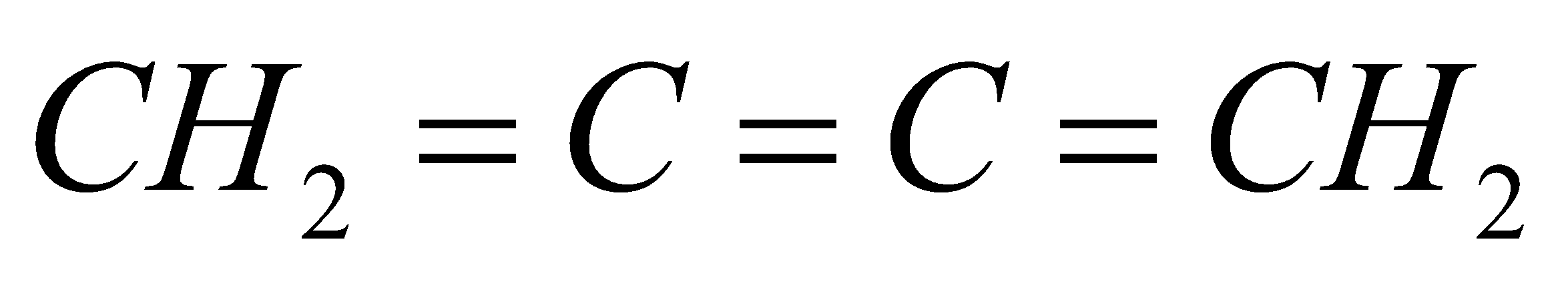
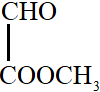


Answer
559.3k+ views
Hint- In order to deal with this question we will use the basic concept of organic chemistry which states that when a carbon atom is $s{p^2}$ or $sp$ hybridized, all the atoms attached to it are in a plane. In this question we will proceed further by understanding when carbon is said to be $sp/s{p^2}/s{p^3}$.
Complete answer:
$sp$ : The third possible arrangement for carbon is sp hybridization which occurs when carbon is bound to two other atoms (two double bonds or one single + one triple bond).
$s{p^2}$ : carbon is said to be $s{p^2}$ hybridized when it is DOUBLY BONDED with any 1 atom and SINGLY BONDED with any other 2 atoms.
$s{p^3}$ : The term “ $s{p^3}$ hybridization” refers to the mixing character of one 2s-orbitals and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be $s{p^3}$ hybridized, it must have an s orbital and three p orbitals
We know that when a carbon atom is $s{p^2}$ or $sp$ hybridized, all the atoms attached to it are in a plane.
All the carbon atoms in compounds A, C and D are $s{p^2}$ hybridized. Hence, in these molecules, all the atoms are coplanar.
In option B, one carbon atom is $s{p^3}$ hybridized. It has tetrahedral geometry. Hence, in this molecule, all atoms are not coplanar.
So, the correct answer is option B.
Note- Coplanar means atoms or groups of atoms that lie on the same plane. Like biphenyl, it has both benzene rings on the same plane. When all atoms of a compound are in the same plane are called coplanar compounds. Co planarity in organic compounds is seen in unsaturated molecules.
Complete answer:
$sp$ : The third possible arrangement for carbon is sp hybridization which occurs when carbon is bound to two other atoms (two double bonds or one single + one triple bond).
$s{p^2}$ : carbon is said to be $s{p^2}$ hybridized when it is DOUBLY BONDED with any 1 atom and SINGLY BONDED with any other 2 atoms.
$s{p^3}$ : The term “ $s{p^3}$ hybridization” refers to the mixing character of one 2s-orbitals and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be $s{p^3}$ hybridized, it must have an s orbital and three p orbitals
We know that when a carbon atom is $s{p^2}$ or $sp$ hybridized, all the atoms attached to it are in a plane.
All the carbon atoms in compounds A, C and D are $s{p^2}$ hybridized. Hence, in these molecules, all the atoms are coplanar.
In option B, one carbon atom is $s{p^3}$ hybridized. It has tetrahedral geometry. Hence, in this molecule, all atoms are not coplanar.
So, the correct answer is option B.
Note- Coplanar means atoms or groups of atoms that lie on the same plane. Like biphenyl, it has both benzene rings on the same plane. When all atoms of a compound are in the same plane are called coplanar compounds. Co planarity in organic compounds is seen in unsaturated molecules.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




