In the structure given below, how many heteroatoms are present?


Answer
593.4k+ views
Hint:
Heteroatoms are defined as the atoms other than carbon and hydrogen in a given compound. There are limited number of hetero atom in a compound. In a compound there cannot be more than three heteroatoms.
Complete answer:
-Heteroatoms are non hydrogen or non carbon atoms that have replaced carbon or hydrogen in the molecular structure.
-These atoms will have lesser or greater attraction for the electrons than the carbon atom.
-The bond that is present between carbon and the heteroatom is polar in nature.
-Heteroatoms affect the reactivity of the organic molecule.
-The examples of heteroatoms are: oxygen, nitrogen, bromine, chlorine, sulphur, phosphorus, iodine, lithium and magnesium.
-In the diagram given below:
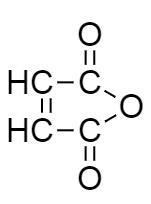
There is only one heteroatom present in a compound and that is oxygen.
Therefore, the compound given in the diagram consists of only one heteroatom.
Additional information:
Example of a compound which consists of two heteroatoms and that is acetamide.
The structure of acetamide is given below as follows:
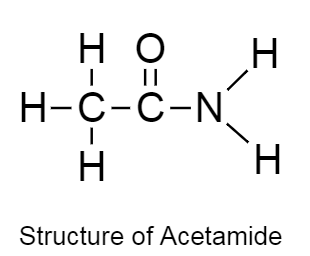
In the given structure there are two heteroatoms present namely: oxygen and nitrogen.
Likewise , we can identify heteroatoms on the basis of the molecular formula also.
Note:The polarity between carbon and oxygen bonds depends on the electron attracting properties of these atoms. For example if there are 2 oxygen atoms in a compound, it will still be considered as one heteroatom because we are only supposed to know which atom is there and not the number of the same heteroatom in a compound.
Heteroatoms are defined as the atoms other than carbon and hydrogen in a given compound. There are limited number of hetero atom in a compound. In a compound there cannot be more than three heteroatoms.
Complete answer:
-Heteroatoms are non hydrogen or non carbon atoms that have replaced carbon or hydrogen in the molecular structure.
-These atoms will have lesser or greater attraction for the electrons than the carbon atom.
-The bond that is present between carbon and the heteroatom is polar in nature.
-Heteroatoms affect the reactivity of the organic molecule.
-The examples of heteroatoms are: oxygen, nitrogen, bromine, chlorine, sulphur, phosphorus, iodine, lithium and magnesium.
-In the diagram given below:

There is only one heteroatom present in a compound and that is oxygen.
Therefore, the compound given in the diagram consists of only one heteroatom.
Additional information:
Example of a compound which consists of two heteroatoms and that is acetamide.
The structure of acetamide is given below as follows:

In the given structure there are two heteroatoms present namely: oxygen and nitrogen.
Likewise , we can identify heteroatoms on the basis of the molecular formula also.
Note:The polarity between carbon and oxygen bonds depends on the electron attracting properties of these atoms. For example if there are 2 oxygen atoms in a compound, it will still be considered as one heteroatom because we are only supposed to know which atom is there and not the number of the same heteroatom in a compound.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

Why is there a time difference of about 5 hours between class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE




