In ${{\text{P}}_{4}}{{\text{O}}_{10}}$, the number of oxygen atoms attached to each phosphorus atom is:
A. 2
B. 3
C. 4
D. 5
Answer
623.1k+ views
Hint: ${{\text{P}}_{4}}{{\text{O}}_{10}}$ is a molecular formula of pentoxide which consists of 4 molecules of phosphorus and 10 molecules of oxygen. The phosphorus can make a maximum of 5 bonds with other molecules whereas oxygen can make a maximum of 2 bonds with other molecules.
Complete Answer:
-To count the number of bonds between oxygen and phosphorus we first have to draw the structure of phosphorus pentoxide.
-The structure is given below:
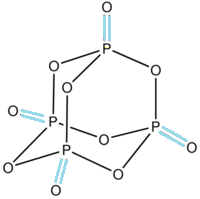
-As we can see in the structure of phosphorus pentoxide there are a total 4 oxygens which are highlighted by blue colour and make double bonds only with a single phosphorus atom.
-The double-bonded oxygen has a hybridisation of $\text{s}{{\text{p}}^{2}}$
-Whereas 6 oxygen atoms are attached to the two phosphorus atoms each with a single bond in which the hybridisation of oxygen atom is $\text{s}{{\text{p}}^{3}}$.
So, option C. is the correct answer.
-The structure of phosphorus pentoxide forms a hexagonal lattice and is held together by the Vander Waal forces.
-The structure consists of at least four polymorphs.
Note: The preparation of phosphorus pentoxide is done when tetraphosphorus reacts with oxygen molecules: ${{\text{P}}_{4}}\text{ + 5}{{\text{O}}_{2}}\text{ }\to \text{ }{{\text{P}}_{4}}{{\text{O}}_{10}}$. The compound is used as a dehydrating agent and is white in appearance. The name of the compound is named so because it is the empirical formula of ${{\text{P}}_{4}}{{\text{O}}_{5}}$.
Complete Answer:
-To count the number of bonds between oxygen and phosphorus we first have to draw the structure of phosphorus pentoxide.
-The structure is given below:

-As we can see in the structure of phosphorus pentoxide there are a total 4 oxygens which are highlighted by blue colour and make double bonds only with a single phosphorus atom.
-The double-bonded oxygen has a hybridisation of $\text{s}{{\text{p}}^{2}}$
-Whereas 6 oxygen atoms are attached to the two phosphorus atoms each with a single bond in which the hybridisation of oxygen atom is $\text{s}{{\text{p}}^{3}}$.
So, option C. is the correct answer.
-The structure of phosphorus pentoxide forms a hexagonal lattice and is held together by the Vander Waal forces.
-The structure consists of at least four polymorphs.
Note: The preparation of phosphorus pentoxide is done when tetraphosphorus reacts with oxygen molecules: ${{\text{P}}_{4}}\text{ + 5}{{\text{O}}_{2}}\text{ }\to \text{ }{{\text{P}}_{4}}{{\text{O}}_{10}}$. The compound is used as a dehydrating agent and is white in appearance. The name of the compound is named so because it is the empirical formula of ${{\text{P}}_{4}}{{\text{O}}_{5}}$.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




