In $N{{O}_{3}}^{-}$ ion, number of bond pair and lone pair of electrons on nitrogen are:
(A) 2,2
(B) 3,1
(C) 1,3
(D) 4,0
Answer
557.9k+ views
Hint: Draw the structure of $N{{O}_{3}}^{-}$ representing all the bonds. Nitrogen cannot have 5 bonds although it shows oxidation state of +5, hence there is a possibility of coordinate bond. The bond pair for every coordinate bond is taken as 1.
Complete step by step answer:
Let us draw the structure of $N{{O}_{3}}^{-}$ to understand the types of bond present in the ion:
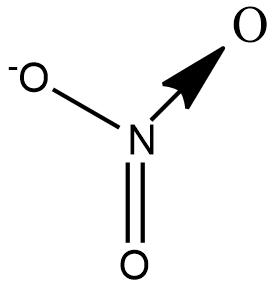
We see that there are 2$\sigma $ bonds, 1$\pi $bond and 1 coordinate bond. Theoretically the coordinate bond is equivalent to a double bond but differs in terms of reactivity towards an incoming ion.
A coordinate bond also called a dative covalent bond is a type of covalent bond in which both electrons come from the same atom. Nitrogen forms a coordinate bond with Oxygen as it cannot have 5 bonds. Although 2 electrons are shared in a coordinate bond, the number of bond pairs is taken to be 1.
Hence the number of bond pairs are 4 and the number of lone pairs is 0.
Therefore, the correct answer is option (D).
Note: Nitrogen shows the oxidation state of +5 in $N{{O}_{3}}^{-}$ ion. However, nitrogen cannot have 5 bonds attached to itself. This is because nitrogen does not have vacant orbitals to expand to its octet. From the subsequent period, the elements have vacant d orbitals and can expand their octet. For e.g. phosphorus can have 5 bonds attached to itself as seen in the molecule $PC{{l}_{5}}$,but $NC{{l}_{5}}$cannot exist.
Complete step by step answer:
Let us draw the structure of $N{{O}_{3}}^{-}$ to understand the types of bond present in the ion:

We see that there are 2$\sigma $ bonds, 1$\pi $bond and 1 coordinate bond. Theoretically the coordinate bond is equivalent to a double bond but differs in terms of reactivity towards an incoming ion.
A coordinate bond also called a dative covalent bond is a type of covalent bond in which both electrons come from the same atom. Nitrogen forms a coordinate bond with Oxygen as it cannot have 5 bonds. Although 2 electrons are shared in a coordinate bond, the number of bond pairs is taken to be 1.
Hence the number of bond pairs are 4 and the number of lone pairs is 0.
Therefore, the correct answer is option (D).
Note: Nitrogen shows the oxidation state of +5 in $N{{O}_{3}}^{-}$ ion. However, nitrogen cannot have 5 bonds attached to itself. This is because nitrogen does not have vacant orbitals to expand to its octet. From the subsequent period, the elements have vacant d orbitals and can expand their octet. For e.g. phosphorus can have 5 bonds attached to itself as seen in the molecule $PC{{l}_{5}}$,but $NC{{l}_{5}}$cannot exist.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




