In an electrolytic cell current flows from?
A.Cathode to anode in outer circuit
B.Anode to cathode outside the cell
C.Cathode to anode inside the cell
D.Anode to cathode inside the cell.
Answer
528.3k+ views
Hint: An electrolytic cell is an electrochemical cell that uses electrical energy to drive a non-spontaneous redox reaction. It is often used to decompose chemical compounds, in a process called electrolysis and Electrons flow from anode to cathode.
Complete answer:
An electrolytic cell converts electrical energy into chemical energy. Here, the redox reaction is spontaneous and is responsible for the production of electrical energy. The reaction at the anode is oxidation and that at the cathode is reduction. The electrons are supplied by the species getting oxidized.
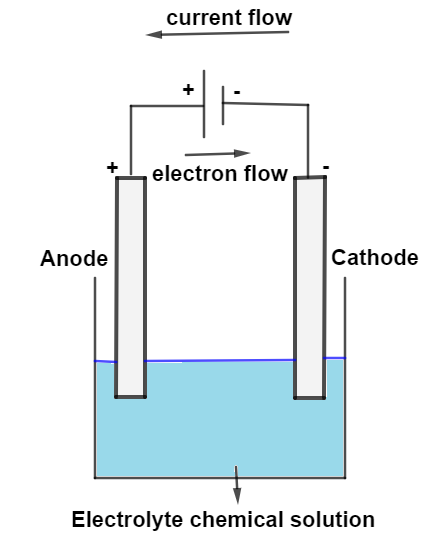
In an electrolytic cell, the negative terminal is the cathode/anode and is the site of the oxidation/reduction half reaction. In an electrolytic cell, the negative terminal is the cathode and is the site of the reduction half reaction. For an electrolytic cell however, this flow is not spontaneous but must be driven by an external power source. In an electrolytic cell, the anode has the "" sign. The potential difference between the electrodes causes electrons to flow from the reductant to the oxidant through the external circuit, generating an electric current.
So, the correct answer is (C) cathode to anode inside the cell.
Note:
Remember Current does not flow out of a battery. Current, a movement of electrons, flows from one part of the battery richer to the other part poorer of them. Batteries put out direct current, as opposed to alternating current, which is what comes out of a wall socket. With direct current, the charge flows only in one direction. With alternating current, the charges slosh back and forth, continually reversing direction.
Complete answer:
An electrolytic cell converts electrical energy into chemical energy. Here, the redox reaction is spontaneous and is responsible for the production of electrical energy. The reaction at the anode is oxidation and that at the cathode is reduction. The electrons are supplied by the species getting oxidized.

In an electrolytic cell, the negative terminal is the cathode/anode and is the site of the oxidation/reduction half reaction. In an electrolytic cell, the negative terminal is the cathode and is the site of the reduction half reaction. For an electrolytic cell however, this flow is not spontaneous but must be driven by an external power source. In an electrolytic cell, the anode has the "" sign. The potential difference between the electrodes causes electrons to flow from the reductant to the oxidant through the external circuit, generating an electric current.
So, the correct answer is (C) cathode to anode inside the cell.
Note:
Remember Current does not flow out of a battery. Current, a movement of electrons, flows from one part of the battery richer to the other part poorer of them. Batteries put out direct current, as opposed to alternating current, which is what comes out of a wall socket. With direct current, the charge flows only in one direction. With alternating current, the charges slosh back and forth, continually reversing direction.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




