In acid catalyzed dehydration of alcohol which of the following alkene is formed.
The reaction may involve rearrangement.
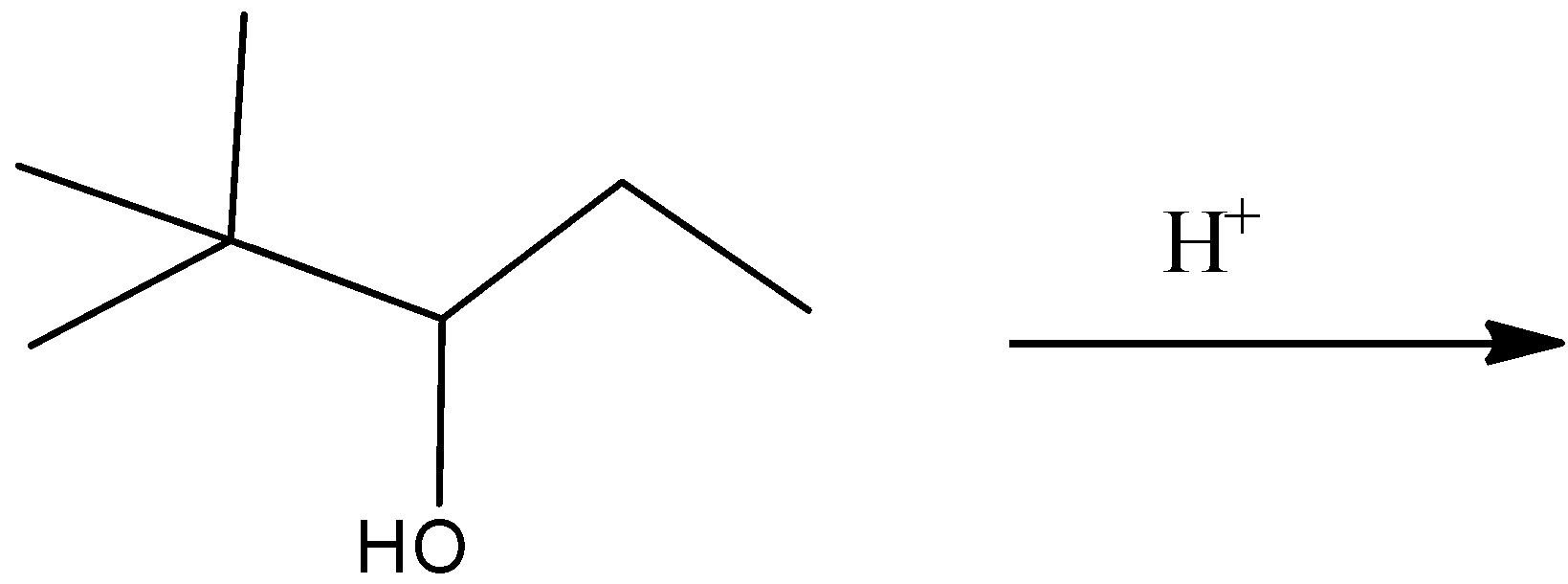
A)
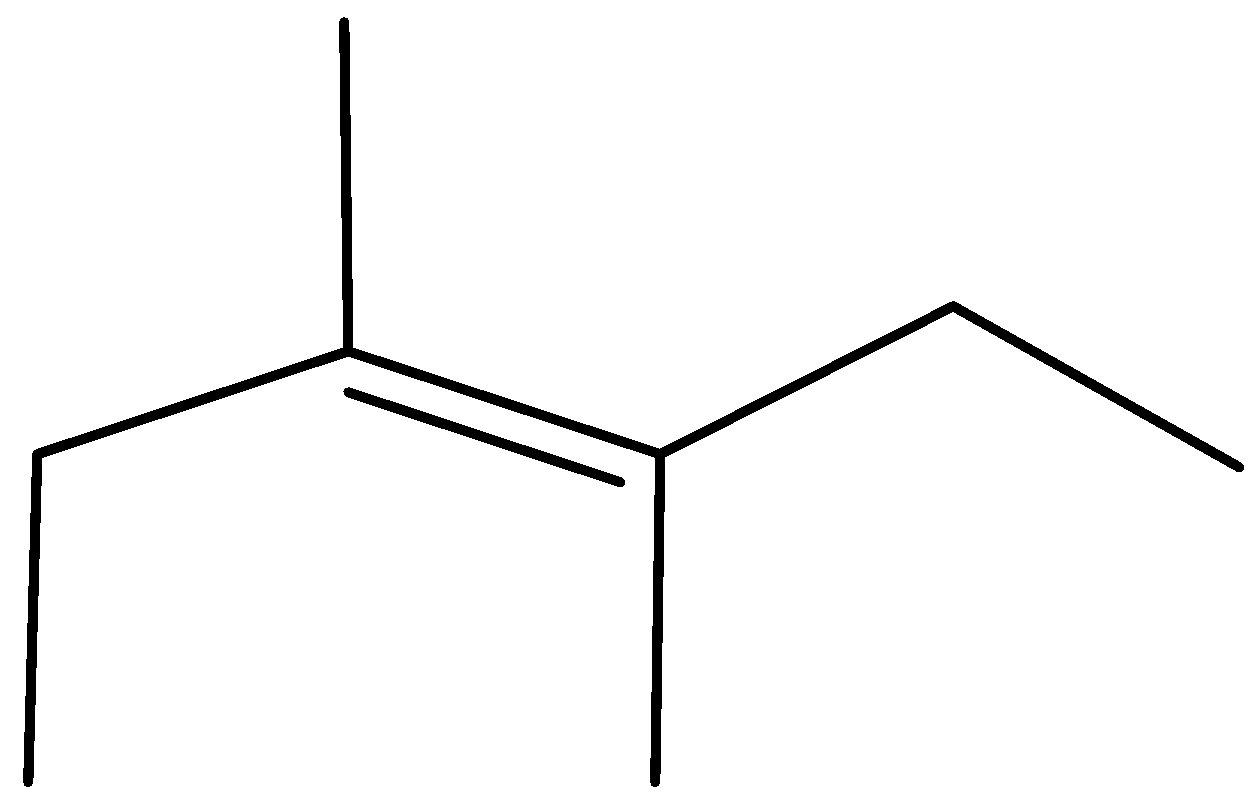
B)
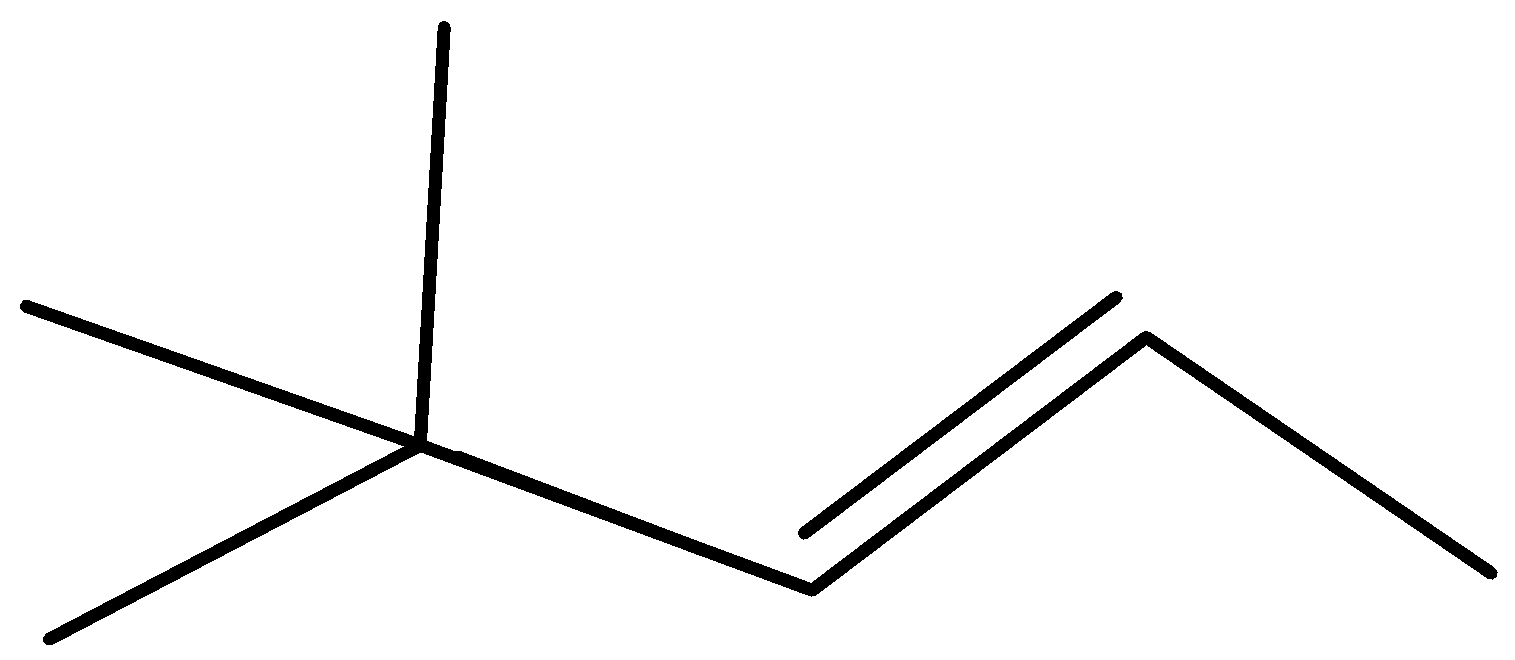
C)
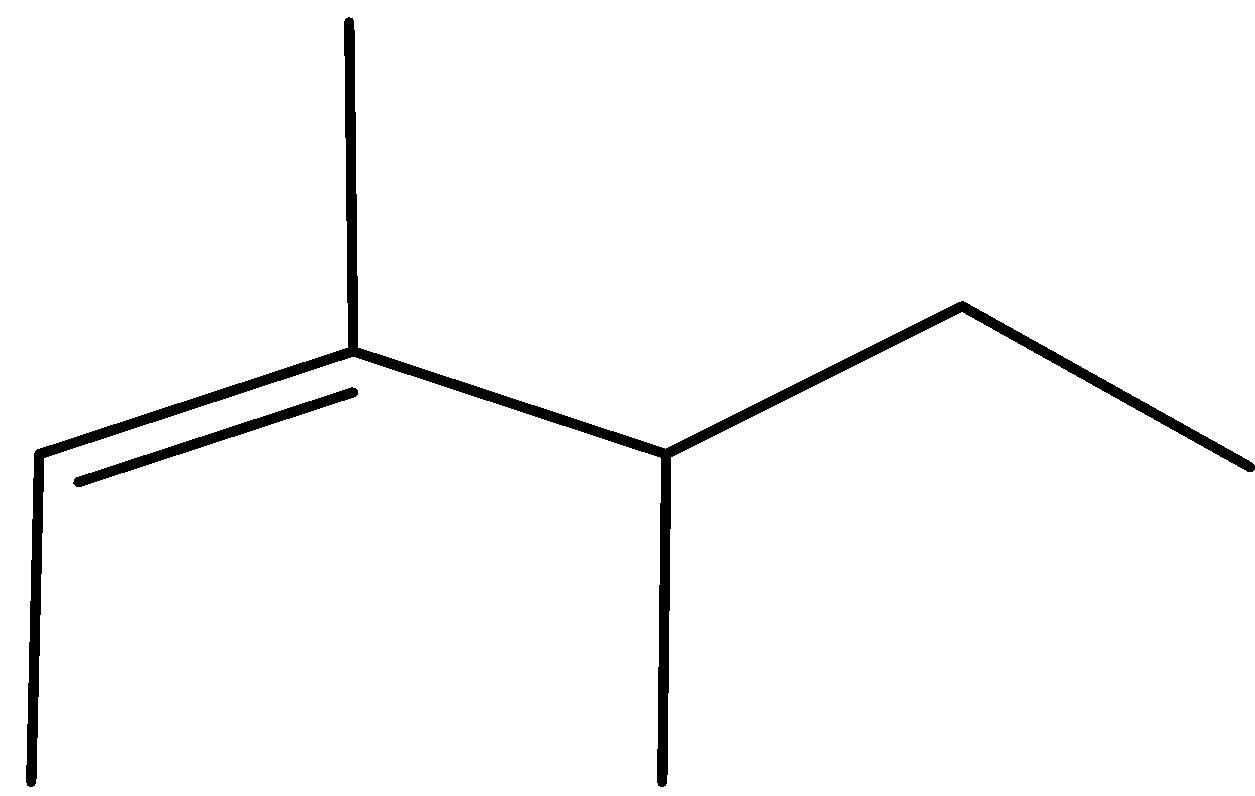
D)
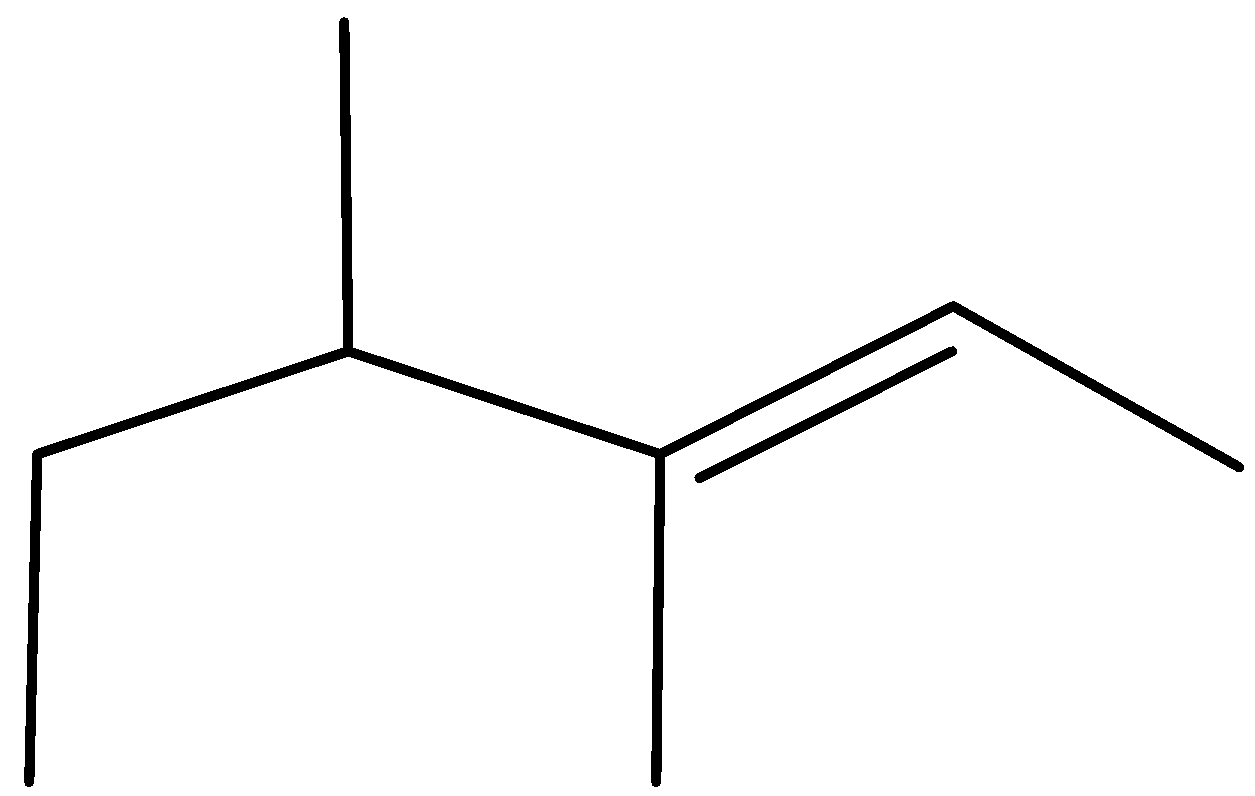





Answer
600.9k+ views
Hint: We must remember that the dehydration of alcohols is a way to synthesize alkenes is by dehydration of alcohols, a process during which alcohols undergo \[E_1{\text{ }}or{\text{ }}E_2\] mechanisms to lose water and form a covalent bond. The dehydration reaction of alcohols to get alkene proceeds by heating the alcohols within the presence of a robust acid, like sulfuric or ortho phosphoric acid, at high temperatures.
Complete step by step answer:As we know that the \[{H^ + }\] attach to OH group due to the presence of lone pair of electron and form \[OH{2^ + }\] which may be a good leaving group so removal of water takes place and carbocation is made and then methyl shift takes place because tertiary carbocation is more stable than secondary carbocation and eventually removal of \[{H^ + }\] ion happen and formation of most stable alkene takes place stability of alkene is defined by the amount of alpha hydrogen present; more the amount of alpha hydrogen more are going to be the stability so the merchandise formed have \[11{\text{ }}\alpha \] hydrogen and hence most stable.
Therefore, Option (A) is correct.
Note:Now we discuss about the nature of carbocation as,
Carbocation:
An ion in which the carbon carries a positive charge is called carbocation and they are formed by the heterolytic cleavage.
Stability of the carbocation:
The stability of the allylic carbocation depends on the inductive effect of the alkyl group attached to the carbocation. The stability of the carbocation increases with an increase in the number of alkyl groups.
The stability order of carbocation is given below,
${{\text{3}}^{\text{o}}}{\text{ > }}{{\text{2}}^{\text{o}}}{\text{ > }}{{\text{1}}^{\text{o}}}$.
Complete step by step answer:As we know that the \[{H^ + }\] attach to OH group due to the presence of lone pair of electron and form \[OH{2^ + }\] which may be a good leaving group so removal of water takes place and carbocation is made and then methyl shift takes place because tertiary carbocation is more stable than secondary carbocation and eventually removal of \[{H^ + }\] ion happen and formation of most stable alkene takes place stability of alkene is defined by the amount of alpha hydrogen present; more the amount of alpha hydrogen more are going to be the stability so the merchandise formed have \[11{\text{ }}\alpha \] hydrogen and hence most stable.
Therefore, Option (A) is correct.
Note:Now we discuss about the nature of carbocation as,
Carbocation:
An ion in which the carbon carries a positive charge is called carbocation and they are formed by the heterolytic cleavage.
Stability of the carbocation:
The stability of the allylic carbocation depends on the inductive effect of the alkyl group attached to the carbocation. The stability of the carbocation increases with an increase in the number of alkyl groups.
The stability order of carbocation is given below,
${{\text{3}}^{\text{o}}}{\text{ > }}{{\text{2}}^{\text{o}}}{\text{ > }}{{\text{1}}^{\text{o}}}$.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




