If ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}} + {\text{NaCN}} \to {\text{A}}\xrightarrow{{{{\text{H}}_{\text{3}}}{\text{O}}}}{\text{B}} + {\text{NaOH}}\xrightarrow{{{\text{CaO}}}}{\text{C}}$
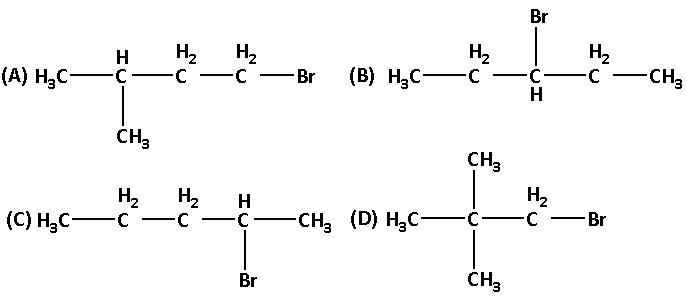
‘C’ has the formula ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$ which can give four structural isomeric monochloro derivatives. What is the structure of ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}}$?

Answer
589.5k+ views
Hint: We are given a reaction sequence. The initial compound is an alkyl halide or alkyl bromide which reacts with sodium cyanide. During the reaction, the cyano groups replace the two bromine atoms. This is a nucleophilic substitution reaction which produces alkyl cyanide. To solve this we have to determine the structure of initial alkyl halide.
Complete step-by-step answer:
We are given a reaction sequence as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}} + {\text{NaCN}} \to {\text{A}}\xrightarrow{{{{\text{H}}_{\text{3}}}{\text{O}}}}{\text{B}} + {\text{NaOH}}\xrightarrow{{{\text{CaO}}}}{\text{C}}$
In the reaction sequence, the initial compound is an alkyl halide or we can say alkyl bromide. The alkyl bromide reacts with sodium cyanide. This is a nucleophilic substitution reaction. During the reaction, the cyano group from sodium cyanide replaces the bromine atom of alkyl bromide. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}} + {\text{NaCN}} \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}}$
Thus, A is alkyl cyanide ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}}$.
The alkyl cyanide then undergoes hydrolysis and produces a carboxylic acid. Alkyl cyanide reacts with ${{\text{H}}_{\text{3}}}{{\text{O}}^ + }$ and produces a carboxylic acid. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}} + {{\text{H}}_{\text{3}}}{{\text{O}}^ + } \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}}$
Thus, B is a carboxylic acid ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}}$.
The carboxylic acid then reacts with sodium hydroxide in presence of calcium oxide. During this reaction first a sodium salt of carboxylic acid is formed which on reaction with calcium oxide loses carbon dioxide molecule. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}} + {\text{NaOH}} \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COONa}}\xrightarrow{{{\text{CaO}}}}{{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$
Thus, C is ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$.
We are given that C can give four structurally isomeric monochloro derivatives. The four structural isomeric monochloro derivatives of C are as follows:
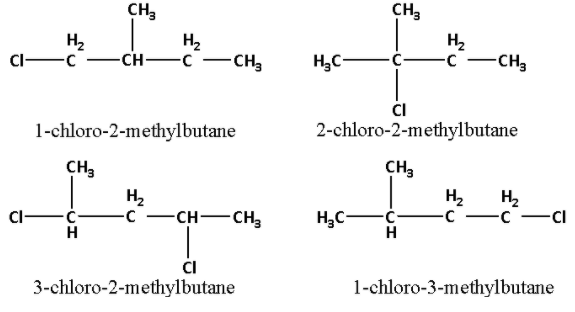
Thus, the structure of ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$ i.e. C is as follows:

The name of compound C is 2-methyl butane.
Thus, compound A will also have the structure in which one methyl group is at carbon number 2.
Hence the correct answer is option ‘A’.
Note: The reaction in which one nucleophile is replaced by another nucleophile is known as a nucleophilic substitution reaction. The group which takes the electron pair with it and gets displaced from the carbon is known as the leaving group. The leaving group leaves as an anion or as a neutral molecule leaving behind a carbonium ion.
Complete step-by-step answer:
We are given a reaction sequence as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}} + {\text{NaCN}} \to {\text{A}}\xrightarrow{{{{\text{H}}_{\text{3}}}{\text{O}}}}{\text{B}} + {\text{NaOH}}\xrightarrow{{{\text{CaO}}}}{\text{C}}$
In the reaction sequence, the initial compound is an alkyl halide or we can say alkyl bromide. The alkyl bromide reacts with sodium cyanide. This is a nucleophilic substitution reaction. During the reaction, the cyano group from sodium cyanide replaces the bromine atom of alkyl bromide. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{Br}} + {\text{NaCN}} \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}}$
Thus, A is alkyl cyanide ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}}$.
The alkyl cyanide then undergoes hydrolysis and produces a carboxylic acid. Alkyl cyanide reacts with ${{\text{H}}_{\text{3}}}{{\text{O}}^ + }$ and produces a carboxylic acid. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{CN}} + {{\text{H}}_{\text{3}}}{{\text{O}}^ + } \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}}$
Thus, B is a carboxylic acid ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}}$.
The carboxylic acid then reacts with sodium hydroxide in presence of calcium oxide. During this reaction first a sodium salt of carboxylic acid is formed which on reaction with calcium oxide loses carbon dioxide molecule. The reaction is as follows:
${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COOH}} + {\text{NaOH}} \to {{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{11}}}}{\text{COONa}}\xrightarrow{{{\text{CaO}}}}{{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$
Thus, C is ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$.
We are given that C can give four structurally isomeric monochloro derivatives. The four structural isomeric monochloro derivatives of C are as follows:

Thus, the structure of ${{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}$ i.e. C is as follows:

The name of compound C is 2-methyl butane.
Thus, compound A will also have the structure in which one methyl group is at carbon number 2.
Hence the correct answer is option ‘A’.
Note: The reaction in which one nucleophile is replaced by another nucleophile is known as a nucleophilic substitution reaction. The group which takes the electron pair with it and gets displaced from the carbon is known as the leaving group. The leaving group leaves as an anion or as a neutral molecule leaving behind a carbonium ion.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE




