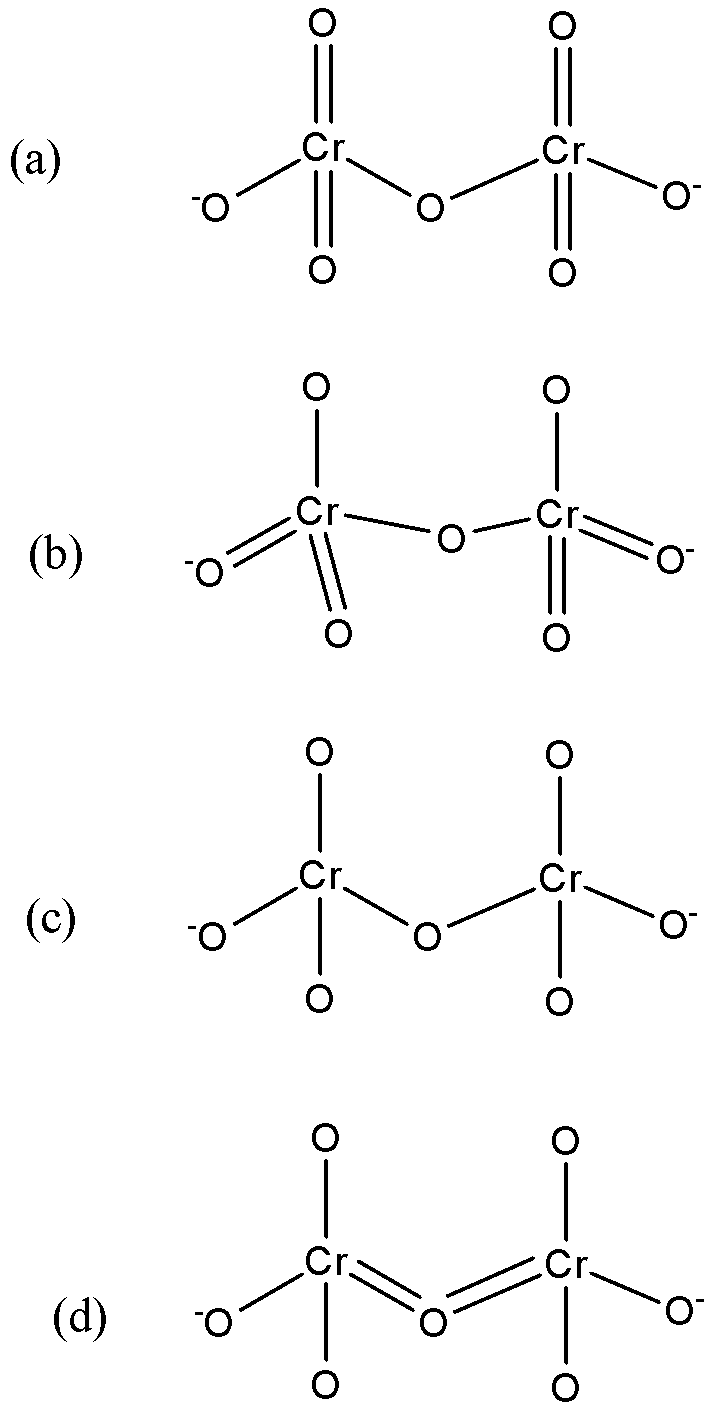
Identify the correct structure of dichromate ion:

Answer
621.3k+ views
Hint: The correct structure has 4 double bonds. All the 4 double bonds are arranged in such a manner that they have the least repulsion and they have symmetry in the structure.
Complete step by step solution:
The formula of dichromate is $C{{r}_{2}}{{O}_{7}}^{2-}$.
The dichromate structure has 2 chromium ions as central metal ions forming a bridge with a common oxygen atom.
It has four double bonds- 2 on each chromium ions in such a manner that they are in the opposite direction.
The structure is:
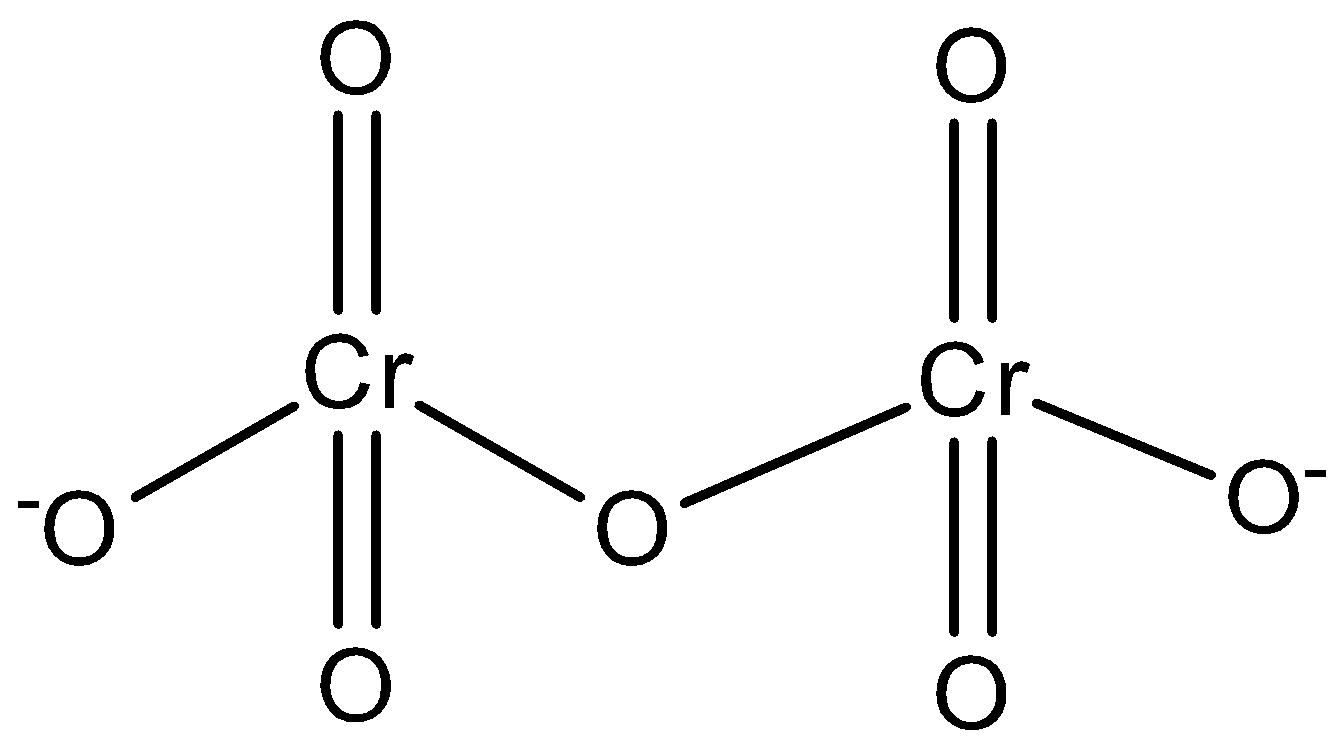
In $C{{r}_{2}}{{O}_{7}}^{2-}$ ion, the two Cr-O bonds which share an oxygen atom at the common vertex of two tetrahedral units are longer than the other six equivalent bonds.
The bond length of Cr and the common O atom is 179pm.
All the other Cr-O bonds are 163pm.
The bond angle of the oxygen atom which is bridged between 2 chromium ions has ${{126}^{\circ }}$.
It is important to know that in both $Cr{{O}_{4}}^{2-}$ and $C{{r}_{2}}{{O}_{7}}^{2-}$ and ions, Cr (VI) has ${{d}^{0}}$a configuration. Hence, the yellow color of $Cr{{O}_{4}}^{2-}$ the orange color $C{{r}_{2}}{{O}_{7}}^{2-}$ is not due to d-d transition but due to charge transfer, i.e., momentary transfer of charge from O-atom to metal atom thereby changing ${{O}^{2-}}$ ion momentarily to ${{O}^{-}}$ ion and reducing the oxidation state of chromium atom from +6 to +5.
So, the correct answer is “Option A”.
Note: You might get confused between the option (a) and (c) because the structure of (c) is also referred but the difference is that in option (c) the 2- charge is on the whole molecule. But the actual structure is an option (a).
Complete step by step solution:
The formula of dichromate is $C{{r}_{2}}{{O}_{7}}^{2-}$.
The dichromate structure has 2 chromium ions as central metal ions forming a bridge with a common oxygen atom.
It has four double bonds- 2 on each chromium ions in such a manner that they are in the opposite direction.
The structure is:

In $C{{r}_{2}}{{O}_{7}}^{2-}$ ion, the two Cr-O bonds which share an oxygen atom at the common vertex of two tetrahedral units are longer than the other six equivalent bonds.
The bond length of Cr and the common O atom is 179pm.
All the other Cr-O bonds are 163pm.
The bond angle of the oxygen atom which is bridged between 2 chromium ions has ${{126}^{\circ }}$.
It is important to know that in both $Cr{{O}_{4}}^{2-}$ and $C{{r}_{2}}{{O}_{7}}^{2-}$ and ions, Cr (VI) has ${{d}^{0}}$a configuration. Hence, the yellow color of $Cr{{O}_{4}}^{2-}$ the orange color $C{{r}_{2}}{{O}_{7}}^{2-}$ is not due to d-d transition but due to charge transfer, i.e., momentary transfer of charge from O-atom to metal atom thereby changing ${{O}^{2-}}$ ion momentarily to ${{O}^{-}}$ ion and reducing the oxidation state of chromium atom from +6 to +5.
So, the correct answer is “Option A”.
Note: You might get confused between the option (a) and (c) because the structure of (c) is also referred but the difference is that in option (c) the 2- charge is on the whole molecule. But the actual structure is an option (a).
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




