How can I draw pi bonds?
Answer
558.9k+ views
Hint: If the p – orbital of one atom is going to overlap with the p – orbital of another atom laterally or side wise then the formed bond in between the two atoms is called $\pi $ - bond or pi -bond. If p – orbital of one atom is going to overlap with the p – orbital of another atom axially then the formed bond in between the two atoms is called $\pi $ - bond
Complete answer:
- In the question it is asked how to draw pi-bond.
- If two s – orbitals of different atoms are going to overlap axially then there is a chance to form a sigma bond ($\sigma $ ).
- If one s – orbital of one atom is going to overlap with p – orbital of another atom axially then also there is a chance to form a sigma bond ($\sigma $ ).
- If two p – orbitals of different atoms are going to overlap sideways then only there is a chance of formation of the pi – bond or $\pi $ -bond.
- The pi-bond can be represented in the form of an image as follows.

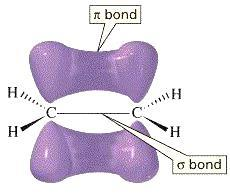
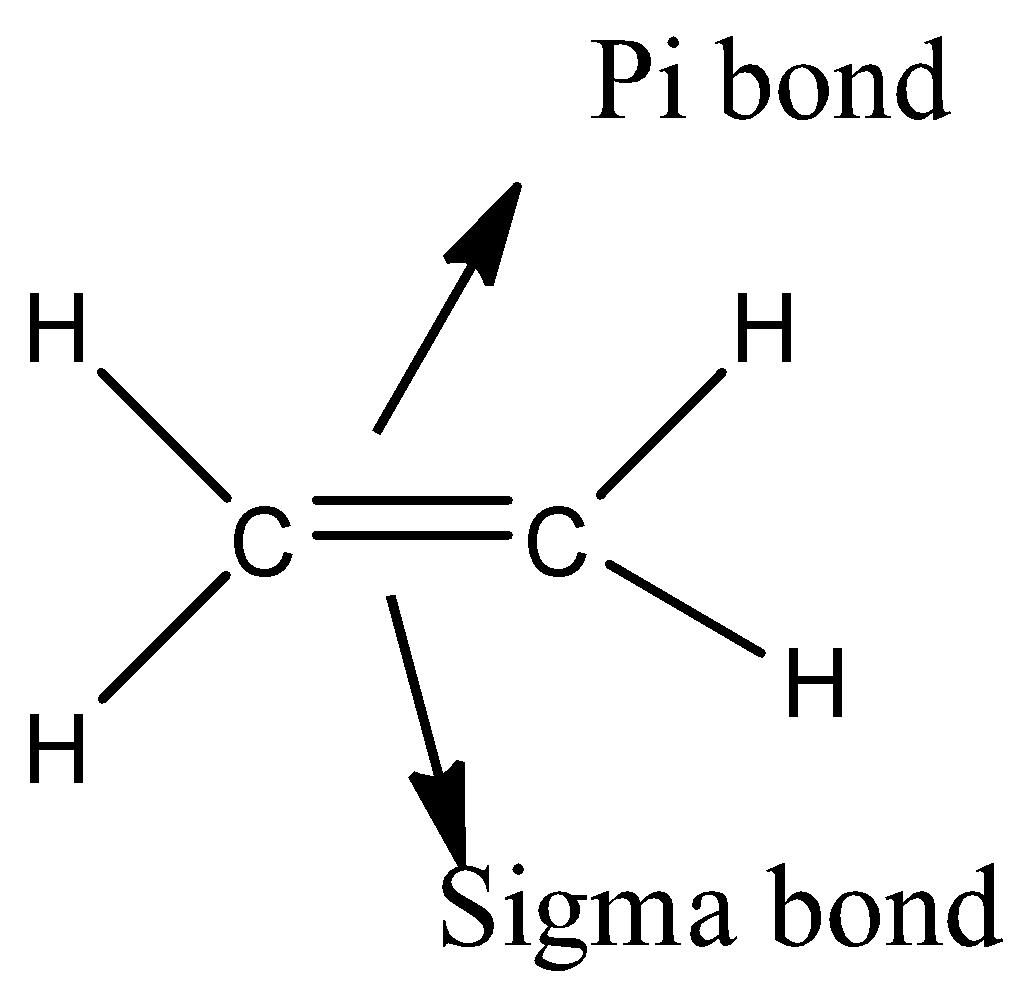
- The molecule which has both a sigma bond and a pi- bond is as follows.
Note:
If a molecule contains one pi-bond means it contains two bonds (One sigma bond and one pi-bond). Double bond is more stable than a single bond because the amount of energy required to break a double bond is more than the amount of energy required to break a single bond.
Complete answer:
- In the question it is asked how to draw pi-bond.
- If two s – orbitals of different atoms are going to overlap axially then there is a chance to form a sigma bond ($\sigma $ ).
- If one s – orbital of one atom is going to overlap with p – orbital of another atom axially then also there is a chance to form a sigma bond ($\sigma $ ).
- If two p – orbitals of different atoms are going to overlap sideways then only there is a chance of formation of the pi – bond or $\pi $ -bond.
- The pi-bond can be represented in the form of an image as follows.


- The molecule which has both a sigma bond and a pi- bond is as follows.

Note:
If a molecule contains one pi-bond means it contains two bonds (One sigma bond and one pi-bond). Double bond is more stable than a single bond because the amount of energy required to break a double bond is more than the amount of energy required to break a single bond.
Recently Updated Pages
The magnetic field in a plane electromagnetic wave class 11 physics CBSE

In a plane electromagnetic wave the electric field class 12 physics CBSE

A plane electromagnetic wave travels in vacuum along class 12 physics CBSE

Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

What are the examples of C3 and C4 plants class 11 biology CBSE

State and prove Bernoullis theorem class 11 physics CBSE

10 examples of friction in our daily life

A body is said to be in dynamic equilibrium if A When class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells




