What happens when
(a) Sodium phenoxide is treated with\[C{H_3}Cl\]?
(b) \[C{H_2} = CH - C{H_2} - OH\] is oxidized by PCC
(c) Phenol is treated with \[C{H_3}COCl\]/anhydrous \[AlC{l_3}\]
Write the chemical equations to support your answer.
Answer
502.5k+ views
Hint: All the above reactions are the direct reactions taking place and all of them are one-step reactions. The first reaction is an example of Williamsons synthesis reaction, the second one is the basic oxidation reaction and the third one is the friedel-craft reaction. By the names of the reactions you can guess what might be the products of these reactions.
Complete step by step answer:
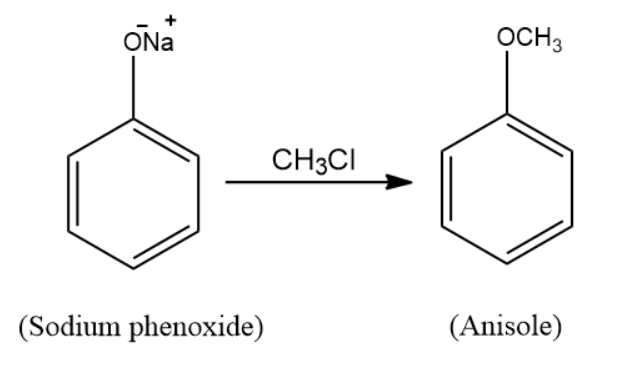
(a) When sodium phenoxide is treated with methyl chloride \[\left( {C{H_3}Cl} \right)\], the product formed is anisole. The reaction taking place here is a nucleophilic substitution reaction. Sodium phenoxide is an alkoxide and methyl chloride is a halide. When the two react, Williamson’s ether synthesis process occurs and the resulting product is ether, which is anisole. The reaction for the following takes place in the following manner:
(b) \[C{H_2} = CH - C{H_2} - OH\] is a primary alcohol. When a primary alcohol is treated with PCC, it undergoes oxidation and gets oxidized to an aldehyde. PCC is Pyridinium chlorochromate (PCC), a complex of chromium trioxide with pyridine and HCl. It is a better reagent for the oxidation of primary alcohols to an aldehyde. The reaction of \[C{H_2} = CH - C{H_2} - OH\] with PCC takes place in the following manner:
$C{H_2} = CH - CH - OH\xrightarrow{{PCC}}C{H_2} = CH - CHO$
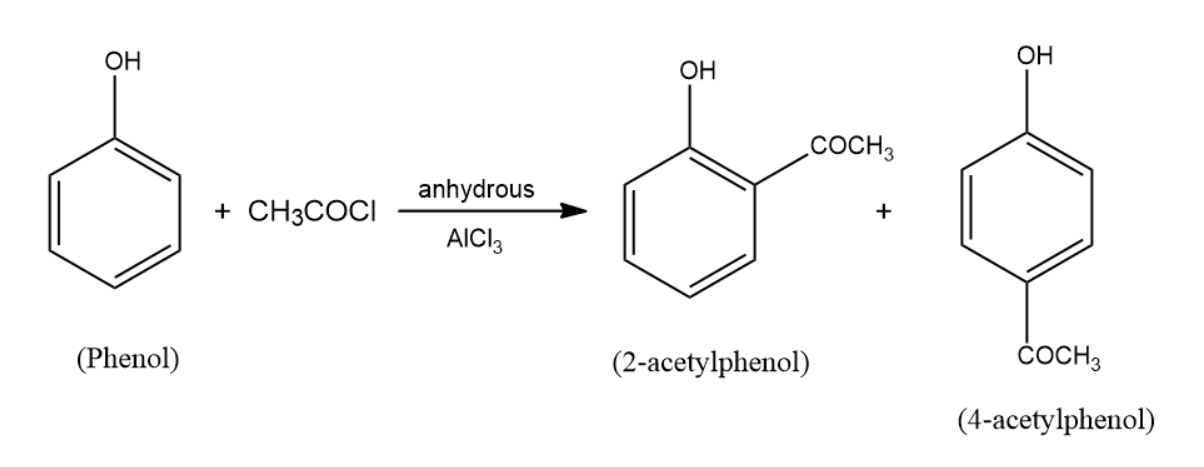
(c) When phenol is treated with \[C{H_3}COCl\]/anhydrous \[AlC{l_3}\] two products are formed among which one of them is major and the other one is minor. The reaction that occurs here is the friedel-craft acylation reaction. The products formed as a result are the two acetylphenol products. These products are $2 - $acetylphenol and $4 - $acetylphenol, among them $4 - $acetylphenol is the major product. The reaction takes place in the following manner:
Note:
Williamson’s synthesis reaction occurs between an organohalide and a deprotonated alcohol to yield ether as the product. This deprotonated alcohol is also called alkoxide. And the reaction occurs via the $Sn2$ mechanism. PCC are very good reagents for converting a primary alcohol to an aldehyde or a secondary alcohol to a ketone. However, PCC cannot oxidize aldehydes to carboxylic acids. Friedel-crafts reactions are a set of reactions developed to attach substituents to an aromatic ring. There is also friedel-craft alkylation in which instead of $C{H_3}COCl$, $C{H_3}Cl$ is used.
Complete step by step answer:
(a) When sodium phenoxide is treated with methyl chloride \[\left( {C{H_3}Cl} \right)\], the product formed is anisole. The reaction taking place here is a nucleophilic substitution reaction. Sodium phenoxide is an alkoxide and methyl chloride is a halide. When the two react, Williamson’s ether synthesis process occurs and the resulting product is ether, which is anisole. The reaction for the following takes place in the following manner:

(b) \[C{H_2} = CH - C{H_2} - OH\] is a primary alcohol. When a primary alcohol is treated with PCC, it undergoes oxidation and gets oxidized to an aldehyde. PCC is Pyridinium chlorochromate (PCC), a complex of chromium trioxide with pyridine and HCl. It is a better reagent for the oxidation of primary alcohols to an aldehyde. The reaction of \[C{H_2} = CH - C{H_2} - OH\] with PCC takes place in the following manner:
$C{H_2} = CH - CH - OH\xrightarrow{{PCC}}C{H_2} = CH - CHO$
(c) When phenol is treated with \[C{H_3}COCl\]/anhydrous \[AlC{l_3}\] two products are formed among which one of them is major and the other one is minor. The reaction that occurs here is the friedel-craft acylation reaction. The products formed as a result are the two acetylphenol products. These products are $2 - $acetylphenol and $4 - $acetylphenol, among them $4 - $acetylphenol is the major product. The reaction takes place in the following manner:

Note:
Williamson’s synthesis reaction occurs between an organohalide and a deprotonated alcohol to yield ether as the product. This deprotonated alcohol is also called alkoxide. And the reaction occurs via the $Sn2$ mechanism. PCC are very good reagents for converting a primary alcohol to an aldehyde or a secondary alcohol to a ketone. However, PCC cannot oxidize aldehydes to carboxylic acids. Friedel-crafts reactions are a set of reactions developed to attach substituents to an aromatic ring. There is also friedel-craft alkylation in which instead of $C{H_3}COCl$, $C{H_3}Cl$ is used.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Find the value of the expression given below sin 30circ class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Name the Largest and the Smallest Cell in the Human Body ?

Draw a diagram showing the external features of fish class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE




