Give the structural formula for 2 -methyl propane.
Answer
548.1k+ views
Hint: A chemical compound's structural formula is a graphic depiction of the molecular structure (determined by structural chemistry methods) that shows how the atoms could be organised in actual three-dimensional space. The molecule's chemical bonding is often depicted, either directly or indirectly.
Complete answer:
Unlike chemical formulas, which have a limited number of symbols and can only describe a limited number of molecular structures, structural formulas have a more complete geometric description of the molecular structure. Many chemical compounds, for example, occur in various isomeric configurations, each with a different enantiomeric structure but the same chemical formula.
Lewis structures (also known as "Lewis dot structures") are two-dimensional graphical formulas that depict atom connectivity and lone pair or unpaired electrons. This is the most popular notation for small molecules. Each line reflects a single bond's two electrons. Double and triple bonds are represented by two or three parallel lines between pairs of atoms, respectively. Pairs of dots may also be used to reflect bonding pairs. Both non-bonded electrons (paired or unpaired) as well as any formal charges on atoms are also seen.
2 methylpropane
Molecular formula =
Longest chain – Propane
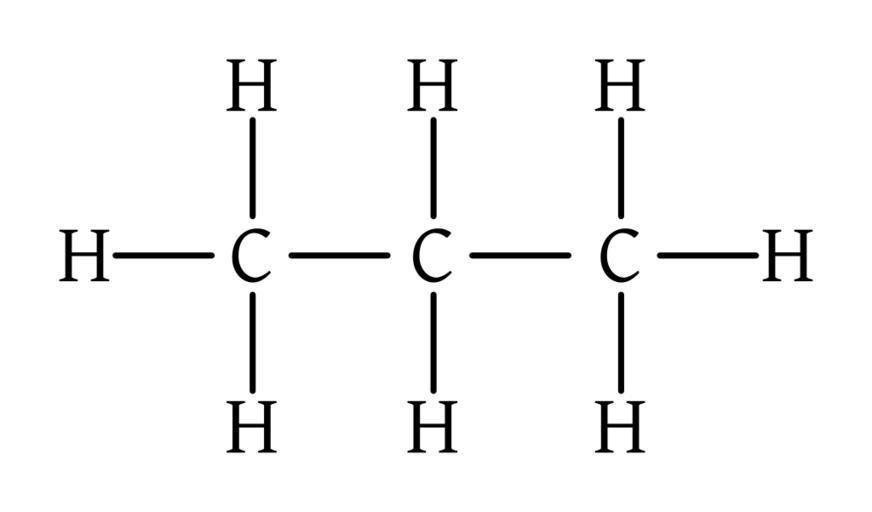
methyl is assigned in the second position
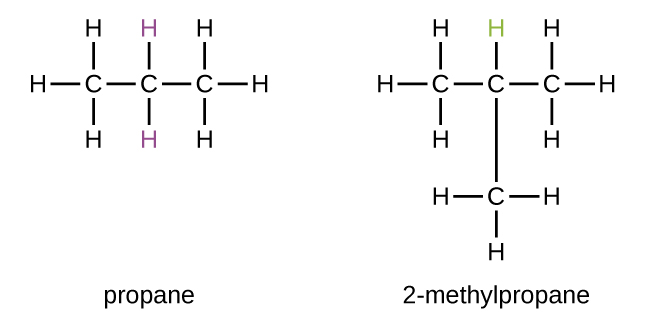
Isobutane is a chemical compound with the molecular formula HC(CH₃)₃. It is also known as i-butane, 2-methylpropane, or methylpropane. It's a kind of butane isomer. Isobutane is a gas that is colourless and odourless. That is the most basic tertiary carbon alkane. In the petrochemical industry, isobutane is used as a precursor molecule, for example, in the synthesis of isooctane.
Note:
Isobutane is the primary feedstock for refinery alkylation units. Gasoline-grade "blendstocks" with high branching for good combustion properties are created using isobutane. 2,4-dimethylpentane and, in particular, 2,2,4-trimethylpentane are typical isobutane drugs.
Complete answer:
Unlike chemical formulas, which have a limited number of symbols and can only describe a limited number of molecular structures, structural formulas have a more complete geometric description of the molecular structure. Many chemical compounds, for example, occur in various isomeric configurations, each with a different enantiomeric structure but the same chemical formula.
Lewis structures (also known as "Lewis dot structures") are two-dimensional graphical formulas that depict atom connectivity and lone pair or unpaired electrons. This is the most popular notation for small molecules. Each line reflects a single bond's two electrons. Double and triple bonds are represented by two or three parallel lines between pairs of atoms, respectively. Pairs of dots may also be used to reflect bonding pairs. Both non-bonded electrons (paired or unpaired) as well as any formal charges on atoms are also seen.
2 methylpropane
Molecular formula =
Longest chain – Propane

methyl is assigned in the second position

Isobutane is a chemical compound with the molecular formula HC(CH₃)₃. It is also known as i-butane, 2-methylpropane, or methylpropane. It's a kind of butane isomer. Isobutane is a gas that is colourless and odourless. That is the most basic tertiary carbon alkane. In the petrochemical industry, isobutane is used as a precursor molecule, for example, in the synthesis of isooctane.
Note:
Isobutane is the primary feedstock for refinery alkylation units. Gasoline-grade "blendstocks" with high branching for good combustion properties are created using isobutane. 2,4-dimethylpentane and, in particular, 2,2,4-trimethylpentane are typical isobutane drugs.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




