Give balanced equation for: A displacement reaction involving a metal above hydrogen in the activity series with copper [II] sulphate solution:
Answer
583.8k+ views
Hint: We know that a reaction where displacement of a less reactive metal from its compound occurs by a more reactive metal is termed as displacement reaction. The reactivity of metals is decided by the reactivity series of metals.
Complete step by step answer:
Let’s first understand the activity or reactivity series of metals in detail. This series is a list of metals arranged according to their reactivity. The metals present at the top of the list are more reactive than those present at the bottom. The highly reactive metal displaces less reactive metal in displacement reaction. As potassium is the highest reactive metal it can displace all other metal atoms from their compounds. Similarly, zinc can displace iron from iron sulphate as zinc is more reactive than iron.
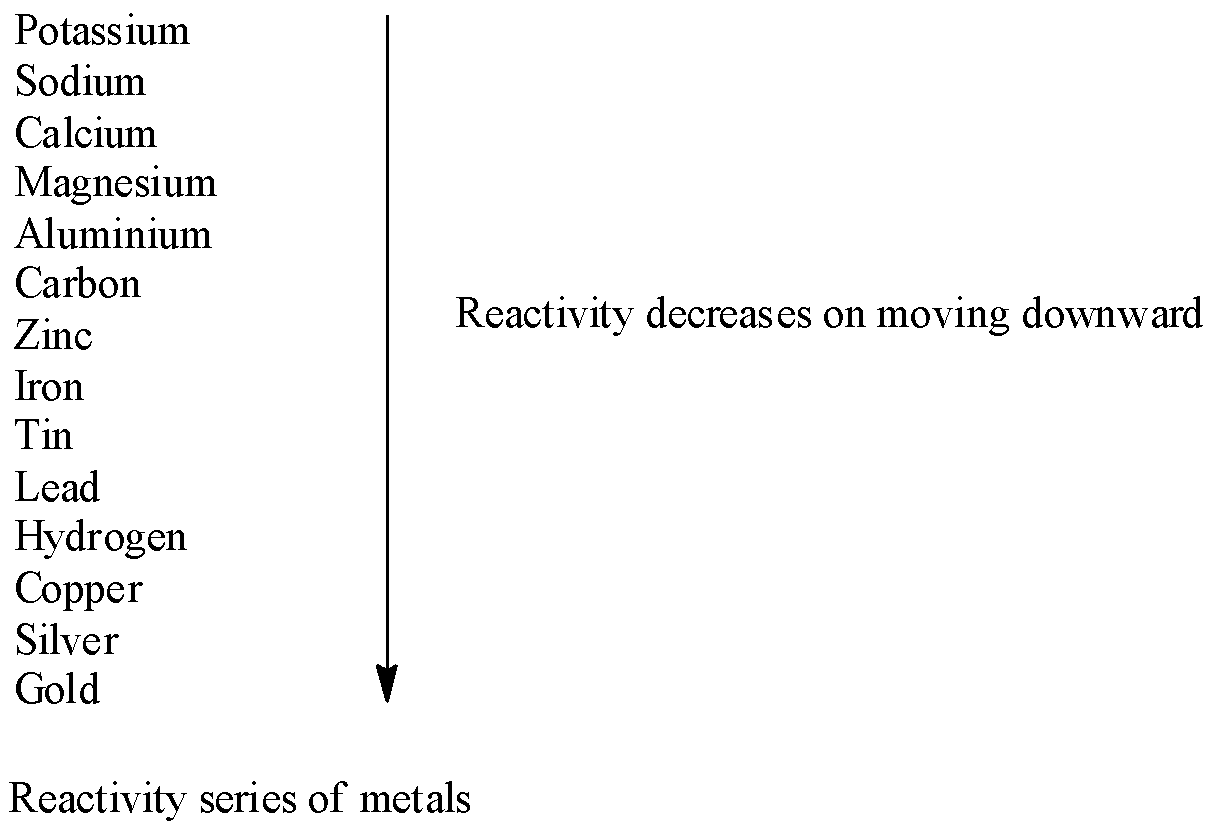
Now come to the question. We have to write a displacement reaction in which a metal above hydrogen in the activity series reacts with copper [II] sulphate solution.
Zinc is the metal which is above hydrogen in the reactivity series. So, we have to write the balanced equation of reaction of zinc and copper sulphate. From the activity series we observe that zinc is more reactive than copper. So, zinc displaces copper from copper sulphate and a displacement reaction takes place.
The balanced equation for the reaction of zinc and copper sulphate is,
${\rm{Zn}} + {\rm{CuS}}{{\rm{O}}_{\rm{4}}} \to {\rm{ZnS}}{{\rm{O}}_{\rm{4}}} + {\rm{Cu}}$
Note: Always remember that in a double displacement reaction, the exchange of ions of two ionic compounds takes place. The reaction between potassium nitrate and aluminium chloride represents a double displacement reaction.
\[{\rm{KN}}{{\rm{O}}_{\rm{3}}} + {\rm{AlC}}{{\rm{l}}_{\rm{3}}} \to {\rm{Al}}{\left( {{\rm{N}}{{\rm{O}}_{\rm{3}}}} \right)_3} + {\rm{KCl}}\]
Complete step by step answer:
Let’s first understand the activity or reactivity series of metals in detail. This series is a list of metals arranged according to their reactivity. The metals present at the top of the list are more reactive than those present at the bottom. The highly reactive metal displaces less reactive metal in displacement reaction. As potassium is the highest reactive metal it can displace all other metal atoms from their compounds. Similarly, zinc can displace iron from iron sulphate as zinc is more reactive than iron.

Now come to the question. We have to write a displacement reaction in which a metal above hydrogen in the activity series reacts with copper [II] sulphate solution.
Zinc is the metal which is above hydrogen in the reactivity series. So, we have to write the balanced equation of reaction of zinc and copper sulphate. From the activity series we observe that zinc is more reactive than copper. So, zinc displaces copper from copper sulphate and a displacement reaction takes place.
The balanced equation for the reaction of zinc and copper sulphate is,
${\rm{Zn}} + {\rm{CuS}}{{\rm{O}}_{\rm{4}}} \to {\rm{ZnS}}{{\rm{O}}_{\rm{4}}} + {\rm{Cu}}$
Note: Always remember that in a double displacement reaction, the exchange of ions of two ionic compounds takes place. The reaction between potassium nitrate and aluminium chloride represents a double displacement reaction.
\[{\rm{KN}}{{\rm{O}}_{\rm{3}}} + {\rm{AlC}}{{\rm{l}}_{\rm{3}}} \to {\rm{Al}}{\left( {{\rm{N}}{{\rm{O}}_{\rm{3}}}} \right)_3} + {\rm{KCl}}\]
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




