What is the formula for the compound iodous acid $\left( HI{{O}_{3}} \right)$?
Answer
552.3k+ views
Hint: The compound as described above i.e. iodous acid is the oxoacid of iodine (iodine oxoacid). In chemistry, iodous acid is commonly known as hypo-iodic acid (conjugate acid of an iodide and an iodine oxoacid).
Complete answer:
Let us study the compound in detail
Iodous acid –
It has the molecular weight of 159.911g/mol having molecular formula as $HI{{O}_{2}}$. The structure of the same is as described below-
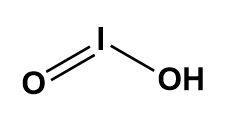
Here, we can see that the number of hydrogen bond donors is one whereas, the number of hydrogen bond acceptors are two.
To note, there are three (worthy) types of acids in the association with iodine;
Iodic acid i.e. $HI{{O}_{3}}$ .
Iodous acid i.e. $HI{{O}_{2}}$ .
Hypo-iodous acid i.e. $HIO$ .
Note:
The oxides of iodine (depending upon the oxidation state of iodine) formed in trace quantities are listed below;
Di-iodine monoxide i.e. ${{I}_{2}}O$ .
Iodine monoxide i.e. $IO$ .
Iodine dioxide i.e. $I{{O}_{2}}$ .
Iodine tetroxide i.e. ${{\left( I{{O}_{2}} \right)}_{2}}$ .
Iodine pentoxide i.e. $\left( O{{\left( O{{I}_{2}} \right)}_{2}} \right)$ .
Complete answer:
Let us study the compound in detail
Iodous acid –
It has the molecular weight of 159.911g/mol having molecular formula as $HI{{O}_{2}}$. The structure of the same is as described below-

Here, we can see that the number of hydrogen bond donors is one whereas, the number of hydrogen bond acceptors are two.
To note, there are three (worthy) types of acids in the association with iodine;
Iodic acid i.e. $HI{{O}_{3}}$ .
Iodous acid i.e. $HI{{O}_{2}}$ .
Hypo-iodous acid i.e. $HIO$ .
Note:
The oxides of iodine (depending upon the oxidation state of iodine) formed in trace quantities are listed below;
Di-iodine monoxide i.e. ${{I}_{2}}O$ .
Iodine monoxide i.e. $IO$ .
Iodine dioxide i.e. $I{{O}_{2}}$ .
Iodine tetroxide i.e. ${{\left( I{{O}_{2}} \right)}_{2}}$ .
Iodine pentoxide i.e. $\left( O{{\left( O{{I}_{2}} \right)}_{2}} \right)$ .
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




