Find packing fraction of three dimensional unit cell of AAAAA……………type hypothetical arrangement in which hexagonal packing is taken in layer.
Answer
597.6k+ views
Hint:To solve this we must first calculate the total area occupied by the spheres in the unit cell and the total area of the square unit cell. The packing efficiency can then be calculated by the ratio of area of spheres in the unit cell to the total area of the square unit cell.
Complete step-by-step answer:
The structure of the hcp unit cell is as follows:
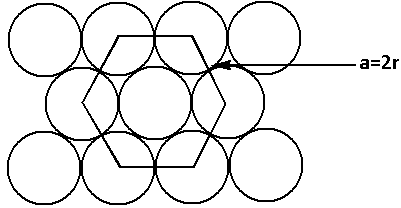
From the structure, we can see that
$a = 2r$ …… (1)
Where $a$ is the side of the face of the hexagon,
$r$ is the radius of the sphere.
We know the formula for the volume of the hexagon,
$V = \dfrac{{3\sqrt 3 }}{2}{a^2} \times h$
Where $V$ is the volume of the hexagon,
$a$ is the side of the face of the hexagon,
$h$ is the height of the hexagon and is equal to $2\sqrt {\dfrac{2}{3}} a$.
Thus, volume of hexagon is,
$V = \dfrac{{3\sqrt 3 }}{2}{a^2} \times 2\sqrt {\dfrac{2}{3}} a$
From equation (1), substitute $a = 2r$. Thus,
$V = \dfrac{{3\sqrt 3 }}{2}{\left( {2r} \right)^2} \times 2\sqrt {\dfrac{2}{3}} 2r$
$V = 24\sqrt 2 {r^3}$ …… (2)
Where $V$ is the volume of the hexagon,
$r$ is the radius of the sphere.
We know the formula for the volume of the sphere is,
$V = \dfrac{4}{3}\pi {r^3}$
Where $V$ is the volume of the sphere,
$r$ is the radius of the sphere.
In a hcp structure, there are a total six spheres. Thus, total volume of the spheres is,
$V = 6\left( {\dfrac{4}{3}\pi {r^3}} \right)$ …… (3)
The packing efficiency is the ratio of volume of spheres in the unit cell to the total volume of the cube cell. Thus, to calculate the packing efficiency divide equation (3) by equation (2). Thus,
${\text{Packing efficiency}} = \dfrac{{6\left( {\dfrac{4}{3}\pi {r^3}} \right)}}{{24\sqrt 2 {r^3}}}$
${\text{Packing efficiency}} = \dfrac{\pi }{{3\sqrt 2 }}$
Substitute $\pi = 3.14$. Thus,
${\text{Packing efficiency}} = \dfrac{{3.14}}{{3\sqrt 2 }}$
${\text{Packing efficiency}} = 0.7402$
Thus, the percentage packing efficiency is $0.7402 \times 100\% = 74.02\% $.
Thus, packaging fraction of three dimensional unit cell of AAAAA……………type hypothetical arrangement in which hexagonal packing is taken in layer is $74\% $.
Note: In hcp structure, each corner atom is shared by six unit cells and thus, the contribution of the corner atoms is two atoms. The tom at the centre is shared by two unit cells and thus, the contribution of the centre atom is one. One single cell contains three atoms. Thus, the total number of atoms in a hcp unit cell are six.
Complete step-by-step answer:
The structure of the hcp unit cell is as follows:

From the structure, we can see that
$a = 2r$ …… (1)
Where $a$ is the side of the face of the hexagon,
$r$ is the radius of the sphere.
We know the formula for the volume of the hexagon,
$V = \dfrac{{3\sqrt 3 }}{2}{a^2} \times h$
Where $V$ is the volume of the hexagon,
$a$ is the side of the face of the hexagon,
$h$ is the height of the hexagon and is equal to $2\sqrt {\dfrac{2}{3}} a$.
Thus, volume of hexagon is,
$V = \dfrac{{3\sqrt 3 }}{2}{a^2} \times 2\sqrt {\dfrac{2}{3}} a$
From equation (1), substitute $a = 2r$. Thus,
$V = \dfrac{{3\sqrt 3 }}{2}{\left( {2r} \right)^2} \times 2\sqrt {\dfrac{2}{3}} 2r$
$V = 24\sqrt 2 {r^3}$ …… (2)
Where $V$ is the volume of the hexagon,
$r$ is the radius of the sphere.
We know the formula for the volume of the sphere is,
$V = \dfrac{4}{3}\pi {r^3}$
Where $V$ is the volume of the sphere,
$r$ is the radius of the sphere.
In a hcp structure, there are a total six spheres. Thus, total volume of the spheres is,
$V = 6\left( {\dfrac{4}{3}\pi {r^3}} \right)$ …… (3)
The packing efficiency is the ratio of volume of spheres in the unit cell to the total volume of the cube cell. Thus, to calculate the packing efficiency divide equation (3) by equation (2). Thus,
${\text{Packing efficiency}} = \dfrac{{6\left( {\dfrac{4}{3}\pi {r^3}} \right)}}{{24\sqrt 2 {r^3}}}$
${\text{Packing efficiency}} = \dfrac{\pi }{{3\sqrt 2 }}$
Substitute $\pi = 3.14$. Thus,
${\text{Packing efficiency}} = \dfrac{{3.14}}{{3\sqrt 2 }}$
${\text{Packing efficiency}} = 0.7402$
Thus, the percentage packing efficiency is $0.7402 \times 100\% = 74.02\% $.
Thus, packaging fraction of three dimensional unit cell of AAAAA……………type hypothetical arrangement in which hexagonal packing is taken in layer is $74\% $.
Note: In hcp structure, each corner atom is shared by six unit cells and thus, the contribution of the corner atoms is two atoms. The tom at the centre is shared by two unit cells and thus, the contribution of the centre atom is one. One single cell contains three atoms. Thus, the total number of atoms in a hcp unit cell are six.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




