Explain with the help of a suitable example polar covalent bond.
Answer
635.4k+ views
Hint: Polar covalent bonding is a type of chemical bond where a pair of electrons are unequally shared between two atoms.
Complete step-by-step answer:
We have been asked to explain polar covalent bonds with a suitable example.
So, let us first discuss the polar covalent bond.
Polar bond-
So, polar covalent bond is a bond which takes place when two different atoms are linked to each other by a covalent bond. In this type of bonding, a chemical bond where a pair of electrons are unequally shared between the atoms. The shared electron pair will not be in the centre just because the bonding atoms differ in electronegativities.
Example of polar covalent bond-
The water molecule shows polar bonding, the water molecule has chemical formula, ${H_2}O$
Now here slightly positive charges are developed in hydrogen atoms and slightly negative charge developed in oxygen atoms as oxygen is more electronegativity than the hydrogen atom. Thus, opposite poles are developed in the molecule.
Refer the figure below to understand the charge distribution-
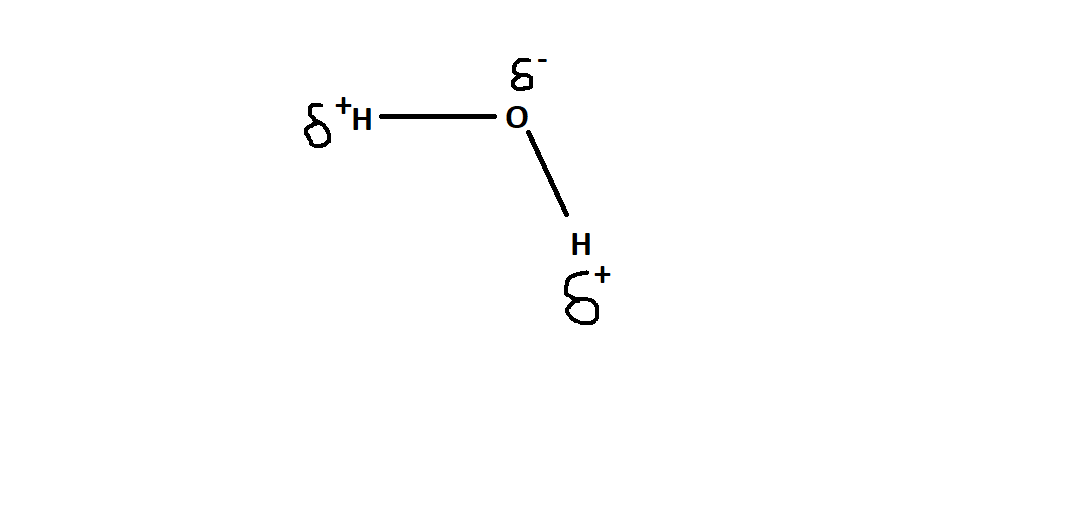
As you can see the above structure has a slight positive charge on hydrogen atoms and a slight negative charge on oxygen atom, hence the bond pair lies towards oxygen atom.
Another example of polar bonding is hydrogen chloride (HCl) molecule. The bonding of hydrogen and chlorine atoms leans more towards Cl atoms because Cl is more electronegative in nature than the hydrogen. Hence, the shared pair of electrons lies not exactly in between the bond but lean towards chlorine. Due to this reason, Cl atoms acquire a slight negative charge, and H atoms acquire slight positive charge.
Refer to the structure below to understand better-
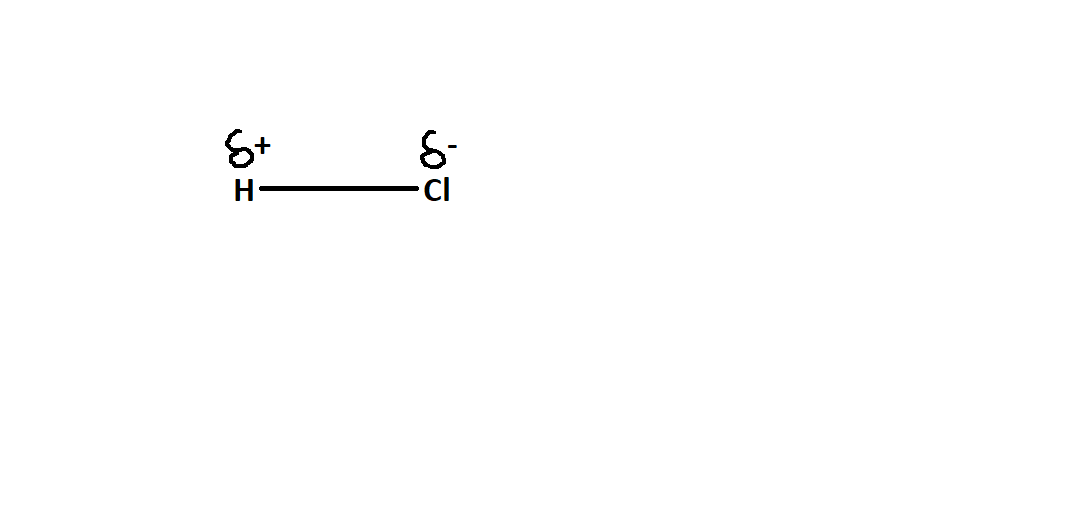 The above structure shows that there is slightly positive charge on hydrogen atom and negative charge on chlorine atom due to its high electronegativity.
The above structure shows that there is slightly positive charge on hydrogen atom and negative charge on chlorine atom due to its high electronegativity.
Note – Whenever such types of questions appear, then first explain what does it mean by polar bonding and what compounds form polar bonds. In polar bonds slightly positive charge appears on the atom which is less electronegative and slightly negative charge appears on the more electronegative atom. As mentioned in the solution, about ${H_2}O$ molecule and $HCl$ molecule and polar bonding is explained with these examples and their structure is also shown.
Complete step-by-step answer:
We have been asked to explain polar covalent bonds with a suitable example.
So, let us first discuss the polar covalent bond.
Polar bond-
So, polar covalent bond is a bond which takes place when two different atoms are linked to each other by a covalent bond. In this type of bonding, a chemical bond where a pair of electrons are unequally shared between the atoms. The shared electron pair will not be in the centre just because the bonding atoms differ in electronegativities.
Example of polar covalent bond-
The water molecule shows polar bonding, the water molecule has chemical formula, ${H_2}O$
Now here slightly positive charges are developed in hydrogen atoms and slightly negative charge developed in oxygen atoms as oxygen is more electronegativity than the hydrogen atom. Thus, opposite poles are developed in the molecule.
Refer the figure below to understand the charge distribution-

As you can see the above structure has a slight positive charge on hydrogen atoms and a slight negative charge on oxygen atom, hence the bond pair lies towards oxygen atom.
Another example of polar bonding is hydrogen chloride (HCl) molecule. The bonding of hydrogen and chlorine atoms leans more towards Cl atoms because Cl is more electronegative in nature than the hydrogen. Hence, the shared pair of electrons lies not exactly in between the bond but lean towards chlorine. Due to this reason, Cl atoms acquire a slight negative charge, and H atoms acquire slight positive charge.
Refer to the structure below to understand better-
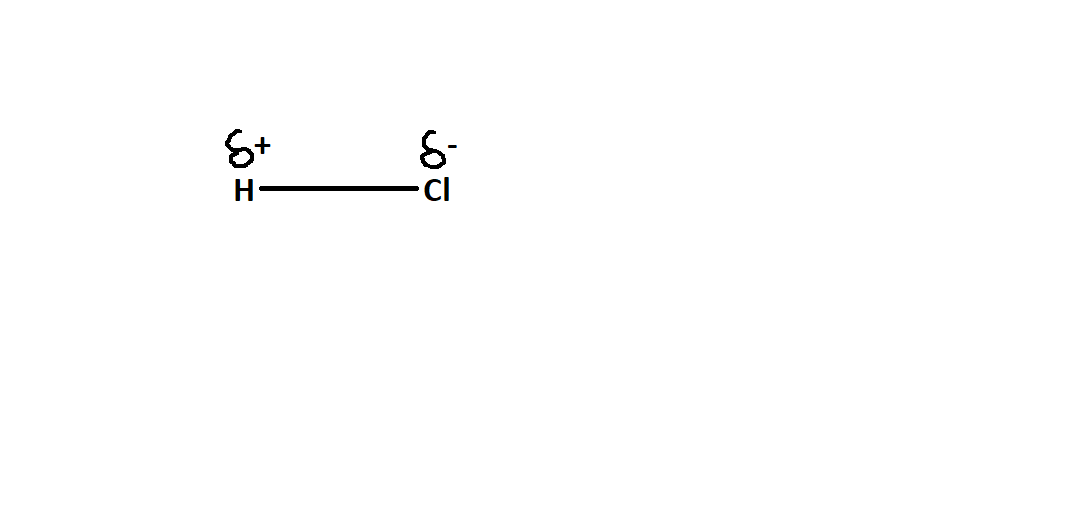
Note – Whenever such types of questions appear, then first explain what does it mean by polar bonding and what compounds form polar bonds. In polar bonds slightly positive charge appears on the atom which is less electronegative and slightly negative charge appears on the more electronegative atom. As mentioned in the solution, about ${H_2}O$ molecule and $HCl$ molecule and polar bonding is explained with these examples and their structure is also shown.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life




