Explain the formation of ${O_2}$ molecule using molecular orbital theory.
Answer
553.1k+ views
Hint: Draw molecular orbital diagram for oxygen molecule. Then, count the number of electrons in bonding molecular orbital and antibonding molecular orbital. After that count the bond order by using the following formula.
Bond Order = $\dfrac{{{\text{Number of electrons in bonding - Number of electrons in antibonding}}}}{2}$
The bond order will tell us the number of bonds that will be formed between two atoms of oxygen molecules.
Complete step by step solution:
The molecular orbital theory is a widely accepted theory for describing the electronic structure of molecules.
The formation of oxygen molecule can be given as –
We know that Oxygen has atomic number = 8. Thus, the electronic configuration for an atom of oxygen in the ground state can be given as –
$1{s^2}2{s^2}2{p^4}$
One atom of oxygen has 8 electrons. Thus, two atoms will possess 16 electrons i.e. Oxygen molecules will have 16 electrons.
The molecular orbital diagram of an Oxygen molecule is as –
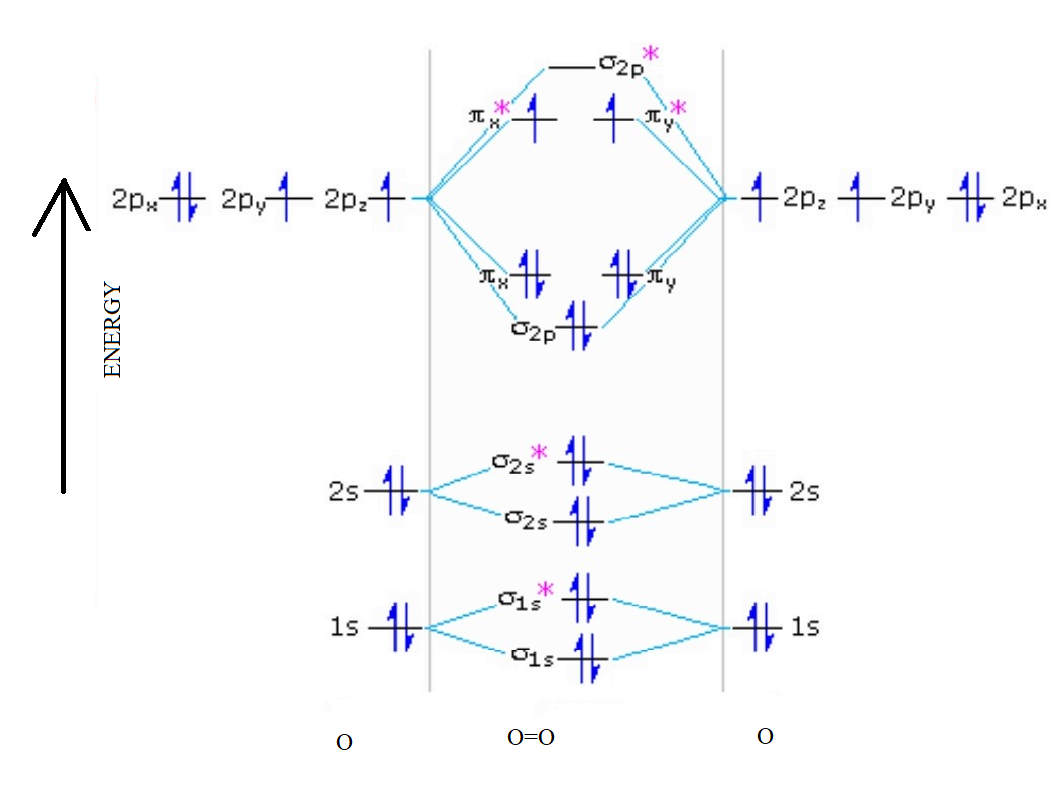
From the diagram, the electronic configuration of oxygen molecule can be written as -
${O_2}$ :- $\sigma 1{s^2}{\sigma ^*}1{s^2}\sigma 2{s^2}{\sigma ^*}2{s^2}\sigma 2{p^2}\Pi 2p_x^2\Pi 2p_y^2{\Pi ^*}2p_x^1{\Pi ^*}2p_y^1$
Now, we can calculate the bond order.
We know the formula for bond order is:-
Bond Order = $\dfrac{{{\text{Number of electrons in bonding - Number of electrons in antibonding}}}}{2}$
Thus, Bond Order = $\dfrac{{10 - 6}}{2}$
Bond Order =2
Thus, the oxygen molecule has two bonds. i.e., one is $\sigma $bond and one $\Pi $ bond.
The two electrons in ${\Pi ^*}2p_x^1$ and ${\Pi ^*}2p_y^1$ orbitals are unpaired. So, the oxygen molecule is paramagnetic in nature due to the presence of these two unpaired electrons.
Note: There are two types of molecular orbital diagrams. The rest of the diagram is the same. The change occurs in two at the point of the splitting of p- orbitals. This should be taken care of so that we choose the right splitting.
Bond Order = $\dfrac{{{\text{Number of electrons in bonding - Number of electrons in antibonding}}}}{2}$
The bond order will tell us the number of bonds that will be formed between two atoms of oxygen molecules.
Complete step by step solution:
The molecular orbital theory is a widely accepted theory for describing the electronic structure of molecules.
The formation of oxygen molecule can be given as –
We know that Oxygen has atomic number = 8. Thus, the electronic configuration for an atom of oxygen in the ground state can be given as –
$1{s^2}2{s^2}2{p^4}$
One atom of oxygen has 8 electrons. Thus, two atoms will possess 16 electrons i.e. Oxygen molecules will have 16 electrons.
The molecular orbital diagram of an Oxygen molecule is as –

From the diagram, the electronic configuration of oxygen molecule can be written as -
${O_2}$ :- $\sigma 1{s^2}{\sigma ^*}1{s^2}\sigma 2{s^2}{\sigma ^*}2{s^2}\sigma 2{p^2}\Pi 2p_x^2\Pi 2p_y^2{\Pi ^*}2p_x^1{\Pi ^*}2p_y^1$
Now, we can calculate the bond order.
We know the formula for bond order is:-
Bond Order = $\dfrac{{{\text{Number of electrons in bonding - Number of electrons in antibonding}}}}{2}$
Thus, Bond Order = $\dfrac{{10 - 6}}{2}$
Bond Order =2
Thus, the oxygen molecule has two bonds. i.e., one is $\sigma $bond and one $\Pi $ bond.
The two electrons in ${\Pi ^*}2p_x^1$ and ${\Pi ^*}2p_y^1$ orbitals are unpaired. So, the oxygen molecule is paramagnetic in nature due to the presence of these two unpaired electrons.
Note: There are two types of molecular orbital diagrams. The rest of the diagram is the same. The change occurs in two at the point of the splitting of p- orbitals. This should be taken care of so that we choose the right splitting.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




