Explain the bonding in methane molecules using electron dot structure.
Answer
596.4k+ views
Hint: The chemical formula of methane molecule is \[{\text{C}}{{\text{H}}_4}\]. It contains four carbon-hydrogen sigma bonds.
Complete answer:
A covalent bond is formed when two atoms share one electron each. Axial overlap of two orbitals form a sigma bond. A covalent sigma bond contains a pair of electrons.
The atomic numbers of hydrogen and carbon are 1 and 6 respectively. The electronic configurations of hydrogen and carbon are \[1{s^1}\] and \[1{s^2}2{s^2}2{p^2}\] respectively. Thus, the number of valence electrons for hydrogen and carbon are 1 and 4 respectively. To attain stability hydrogen should have 2 electrons in its valence shell. For this purpose, a hydrogen atom shares one electron with carbon atom, the carbon atom also shares one electron with this hydrogen atom. Thus, a carbon-hydrogen covalent bond is formed. To attain stability, the carbon atom needs eight electrons in its valence shell. For this purpose, the carbon atom shares 4 electrons with 4 hydrogen atoms to form 4 carbon-hydrogen covalent bonds.
You can write the following structure for methane molecules.
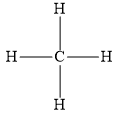
You can write the following electron dot structure for methane molecules.

In the above structure, the dot represents the electron of a carbon atom and the cross represents the electron of hydrogen atom.
Note: Since the central carbon atom in methane molecule has 4 bonds and zero lone pairs of electrons, it will undergo \[s{p^3}\] hybridization to form four degenerate \[s{p^3}\] hybrid orbitals. These four degenerate \[s{p^3}\] hybrid orbitals of carbon overlap with four 1s atomic orbitals of 4 hydrogen atoms to form four carbon-hydrogen covalent bonds. Methane molecules have tetrahedral geometry.
Complete answer:
A covalent bond is formed when two atoms share one electron each. Axial overlap of two orbitals form a sigma bond. A covalent sigma bond contains a pair of electrons.
The atomic numbers of hydrogen and carbon are 1 and 6 respectively. The electronic configurations of hydrogen and carbon are \[1{s^1}\] and \[1{s^2}2{s^2}2{p^2}\] respectively. Thus, the number of valence electrons for hydrogen and carbon are 1 and 4 respectively. To attain stability hydrogen should have 2 electrons in its valence shell. For this purpose, a hydrogen atom shares one electron with carbon atom, the carbon atom also shares one electron with this hydrogen atom. Thus, a carbon-hydrogen covalent bond is formed. To attain stability, the carbon atom needs eight electrons in its valence shell. For this purpose, the carbon atom shares 4 electrons with 4 hydrogen atoms to form 4 carbon-hydrogen covalent bonds.
You can write the following structure for methane molecules.

You can write the following electron dot structure for methane molecules.

In the above structure, the dot represents the electron of a carbon atom and the cross represents the electron of hydrogen atom.
Note: Since the central carbon atom in methane molecule has 4 bonds and zero lone pairs of electrons, it will undergo \[s{p^3}\] hybridization to form four degenerate \[s{p^3}\] hybrid orbitals. These four degenerate \[s{p^3}\] hybrid orbitals of carbon overlap with four 1s atomic orbitals of 4 hydrogen atoms to form four carbon-hydrogen covalent bonds. Methane molecules have tetrahedral geometry.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




