Explain the alkylation of aniline.
Answer
563.7k+ views
Hint: Aniline possesses nitrogen having lone pair of electrons which behaves as a nucleophile. And the reaction of aniline with haloalkanes, which is called alkylation, forms substituted anilines by the mechanism of nucleophilic substitution reaction.
Complete answer:
Aniline belongs to a class of aromatic amine in which $-\text{N}{{\text{H}}_{2}}$ group is directly attached to the aromatic benzene ring system. The nitrogen present in aniline and other aromatic amines is bearing a lone pair of electrons which behaves as a nucleophile and thus aniline undergoes nucleophilic substitution reactions.
Alkylation is the reaction of aniline with haloalkanes which results in the formation of substituted aromatic amine. Aniline reacts with haloalkane and produces a mixture of primary, secondary, tertiary, and quaternary ammonium salt.
The products are formed by the substitution of hydrogen from aniline to an alkyl group. The reaction involved is shown below:
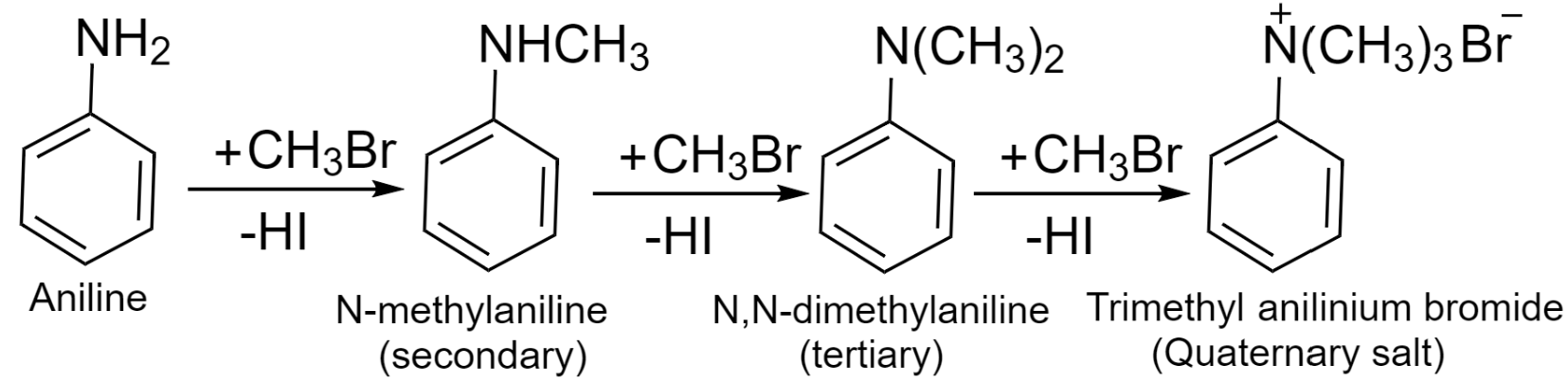
Additional information:
Due to the resonance (+R) effect of $-\text{N}{{\text{H}}_{2}}\text{ , }-\text{NHR , }-\text{N}{{\text{R}}_{2}}$ groups, the electron density at ortho and para positions of benzene ring increases, and thus aromatic amines can also undergo electrophilic substitution reactions.
Note:
The aromatic amines are less basic than the aliphatic amines but more basic than water. On reaction with mineral acids, these compounds form their respective salts. Also, the original amines can be regenerated from their salts by treating them with alkalis.
Complete answer:
Aniline belongs to a class of aromatic amine in which $-\text{N}{{\text{H}}_{2}}$ group is directly attached to the aromatic benzene ring system. The nitrogen present in aniline and other aromatic amines is bearing a lone pair of electrons which behaves as a nucleophile and thus aniline undergoes nucleophilic substitution reactions.
Alkylation is the reaction of aniline with haloalkanes which results in the formation of substituted aromatic amine. Aniline reacts with haloalkane and produces a mixture of primary, secondary, tertiary, and quaternary ammonium salt.
The products are formed by the substitution of hydrogen from aniline to an alkyl group. The reaction involved is shown below:

Additional information:
Due to the resonance (+R) effect of $-\text{N}{{\text{H}}_{2}}\text{ , }-\text{NHR , }-\text{N}{{\text{R}}_{2}}$ groups, the electron density at ortho and para positions of benzene ring increases, and thus aromatic amines can also undergo electrophilic substitution reactions.
Note:
The aromatic amines are less basic than the aliphatic amines but more basic than water. On reaction with mineral acids, these compounds form their respective salts. Also, the original amines can be regenerated from their salts by treating them with alkalis.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




