Explain stereoisomerism in tartaric acid. How many optical isomers are possible for tartaric acid? What are the differences between meso tartaric acid and racemic mixture?
Answer
599.7k+ views
Hint: Stereoisomers are defined as the molecules having the similar bond connectivity but different molecular configuration. Stereoisomers are of two types: optical and geometrical isomers.
Complete Solution :
Compounds which have the same structure but different spatial arrangements are known as stereoisomers and this phenomenon is known as stereoisomerism.
The structure of tartaric acid is mentioned below:
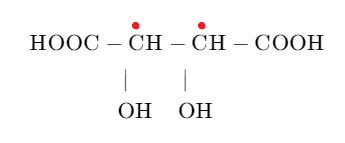
If we analyse the structure, we can see that there are two asymmetric carbon atoms so using the formulae ${{2}^{n}}$ formula where n is the number of asymmetric carbon atoms, we can calculate the number of stereoisomers.
Number of stereoisomers$={{2}^{2}}=4$
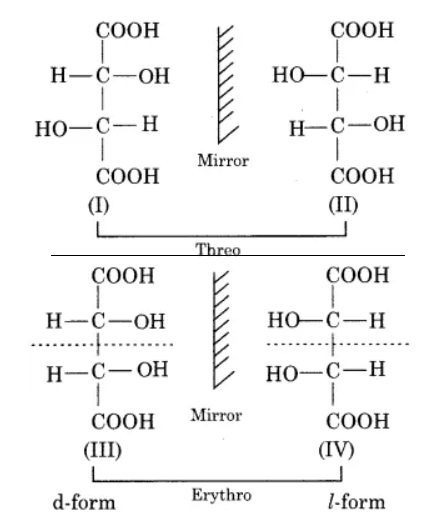
Compounds (I) and (II) are mirror images of each other and they are known as enantiomers but in the meso form. There is a place of symmetry present in tartaric acid. Tartaric acid shows three isomeric forms one of which is optically inactive and the other two are optically active i.e. d (+) and I or (-).
- Difference between meso tartaric acid and racemic mixture is mentioned below:
So, the correct answer is “Option B”.
Note: Meso tartaric acid cannot be separated into two different forms whereas racemic mixture can be separated into two forms.
- To identify the geometrical isomerism we need to restrict the rotation of the carbon-carbon double bond and if there is presence of carbon-carbon double bond then we can check for the possibility of geometrical isomers.
Complete Solution :
Compounds which have the same structure but different spatial arrangements are known as stereoisomers and this phenomenon is known as stereoisomerism.
The structure of tartaric acid is mentioned below:

If we analyse the structure, we can see that there are two asymmetric carbon atoms so using the formulae ${{2}^{n}}$ formula where n is the number of asymmetric carbon atoms, we can calculate the number of stereoisomers.
Number of stereoisomers$={{2}^{2}}=4$

Compounds (I) and (II) are mirror images of each other and they are known as enantiomers but in the meso form. There is a place of symmetry present in tartaric acid. Tartaric acid shows three isomeric forms one of which is optically inactive and the other two are optically active i.e. d (+) and I or (-).
- Difference between meso tartaric acid and racemic mixture is mentioned below:
| Meso tartaric acid | Racemic mixture |
| The angle of optical rotation of meso tartaric acid is 0 degree. | The optical rotation of racemic mixture is also 0 degree. |
| Melting point = 140 degree centigrade. | Melting point = 260 degree centigrade. |
| This is optically inactive due to internal compensation. | This is optically inactive due to external compensation. |
So, the correct answer is “Option B”.
Note: Meso tartaric acid cannot be separated into two different forms whereas racemic mixture can be separated into two forms.
- To identify the geometrical isomerism we need to restrict the rotation of the carbon-carbon double bond and if there is presence of carbon-carbon double bond then we can check for the possibility of geometrical isomers.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




