Explain Friedel-Craft alkylation of chlorobenzene. Give an equation.
Answer
558k+ views
Hint : An alkyl group can be added to a benzene molecule by an electrophilic aromatic substitution reaction called the Friedel‐Crafts alkylation reaction. One example is the addition of a methyl group to a benzene ring. The mechanism for this reaction begins with the generation of a methyl carbocation from methyl bromide.
Complete Step By Step Answer:
Friedel-Crafts Alkylation refers to the replacement of an aromatic proton with an alkyl group. This is done through an electrophilic attack on the aromatic ring with the help of a carbocation. The Friedel-Crafts alkylation reaction is a method of generating alkyl benzenes by using alkyl halides as reactants.
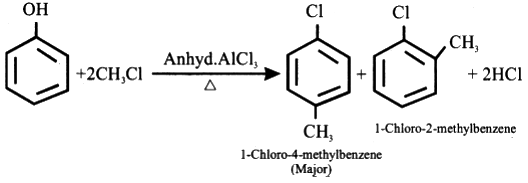
A Lewis acid catalyst such as $ FeC{l_3} $ or $ AlC{l_3} $ is employed in this reaction in order to form a carbocation by facilitating the removal of the halide. The resulting carbocation undergoes a rearrangement before proceeding with the alkylation reaction.
Additional Information:
Mechanism:
The Friedel-Crafts alkylation reaction proceeds via a three-step mechanism.
Step 1
The Lewis acid catalyst ( $ AlC{l_3} $ ) undergoes a reaction with the alkyl halide, resulting in the formation of an electrophilic carbocation.
Step 2
The carbocation proceeds to attack the aromatic ring, forming a cyclohexadienyl cation as an intermediate. The aromaticity of the arene is temporarily lost due to the breakage of the carbon-carbon double bond.
Step 3
The deprotonation of the intermediate leads to the reformation of the carbon-carbon double bond, restoring aromaticity to the compound. This proton goes on to form hydrochloric acid, regenerating the $ AlC{l_3} $ catalyst.
Note :
Friedel craft alkylation will undergo that aromatic compound, which is electron-rich. As it is the electrophilic substitution reaction. But, since the carbo-cations formed by aryl and vinyl halides are extremely unstable, they cannot be used in this reaction. And also the presence of a deactivating group on the aromatic ring can lead to the deactivation of the catalyst due to the formation of complexes.
Complete Step By Step Answer:
Friedel-Crafts Alkylation refers to the replacement of an aromatic proton with an alkyl group. This is done through an electrophilic attack on the aromatic ring with the help of a carbocation. The Friedel-Crafts alkylation reaction is a method of generating alkyl benzenes by using alkyl halides as reactants.

A Lewis acid catalyst such as $ FeC{l_3} $ or $ AlC{l_3} $ is employed in this reaction in order to form a carbocation by facilitating the removal of the halide. The resulting carbocation undergoes a rearrangement before proceeding with the alkylation reaction.
Additional Information:
Mechanism:
The Friedel-Crafts alkylation reaction proceeds via a three-step mechanism.
Step 1
The Lewis acid catalyst ( $ AlC{l_3} $ ) undergoes a reaction with the alkyl halide, resulting in the formation of an electrophilic carbocation.
Step 2
The carbocation proceeds to attack the aromatic ring, forming a cyclohexadienyl cation as an intermediate. The aromaticity of the arene is temporarily lost due to the breakage of the carbon-carbon double bond.
Step 3
The deprotonation of the intermediate leads to the reformation of the carbon-carbon double bond, restoring aromaticity to the compound. This proton goes on to form hydrochloric acid, regenerating the $ AlC{l_3} $ catalyst.
Note :
Friedel craft alkylation will undergo that aromatic compound, which is electron-rich. As it is the electrophilic substitution reaction. But, since the carbo-cations formed by aryl and vinyl halides are extremely unstable, they cannot be used in this reaction. And also the presence of a deactivating group on the aromatic ring can lead to the deactivation of the catalyst due to the formation of complexes.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




