
Enzyme catalyzing optical or geometrical rearrangement of atomic groupings without altering molecular weight or number of atom is
A. Ligase
B. Isomerase
C. Oxidoreductase
D. Hydrolase
Answer
591k+ views
Hint: All living beings have chemical and metabolic reactions occurring within their bodies. These reactions include digestion of food, absorption of molecules, and production of energy. A biochemical reaction is a term used to refer to chemical reactions occurring within the body. Metabolism on the other hand is a combination of all biochemical reactions. Catabolism and anabolism (synthesis) are part of metabolism. Catabolism refers to the breakdown of substances, whereas anabolism refers to the formation of substances.
Step by step answer:The biochemical reactions occurring within the body is catalyzed by globular proteins called enzymes. They only act by catalyzing the chemical reaction and do not undergo any changes themselves. A particular reaction and substrate have a specific enzyme. The enzymes are named based on the reaction or compound they participate in. Enzymes are often termed as biocatalysts. For instance, the hydrolysis of maltose to glucose is catalyzed by the enzyme maltase.
${{C}_{12}}{{H}_{22}}{{O}_{11}}\xrightarrow{maltase}2{{C}_{6}}{{H}_{12}}{{O}_{6}}$
$Maltose\xrightarrow{maltase}Glu\cos e$
-Ligase: Ligase can be defined as an enzyme that can be utilized to catalyze the fusion of two large molecules by the formation of a new chemical compound. This is usually accompanied by hydrolysis. Ligases such as DNA ligase are enzymes that are utilized in molecular biology laboratories where these enzymes assist in recombinant DNA experiments including DNA replication, DNA repair, and joining DNA fragments. This is achieved by the catalyzed formation of a phosphodiester bond.
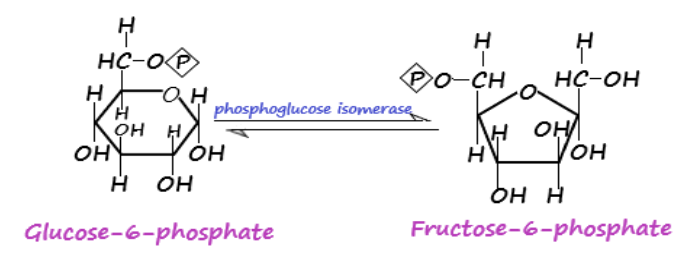
-Isomerase: Enzyme that assists in the conversion of a molecule from one isomer to another with the end product having the same molecular formula but different spatial arrangements are called isomerase. Intramolecular rearrangements involve the breaking and formation of bonds. This is facilitated by isomerases. For instance, the glycolysis process involves the conversion of glucose-6-phosphate to fructose-6-phosphate in presence of phosphoglucose isomerase catalyst.
-Oxidoreductase: Transfer of electrons from the reductant to the oxidant is catalyzed by the enzyme oxidoreductase. The reductant is also known as an electron donor, while oxidant is referred to as the electron acceptor. Oxidoreductase enzymes utilize cofactors such as NADP or NAD+.
-Hydrolase: Hydrolase is a biochemical catalyst designed to break a chemical bond through hydrolysis. Hydrolysis is defined as the addition of a water molecule to a chemical process. This results in the breakdown of larger molecules to smaller compounds or molecules.
Therefore the correct option is B, i.e., Isomerase
Note:For the progress of a chemical or biochemical reaction, only a small amount of enzyme is required. Enzymes are believed to decrease the activation energy’s magnitude. For instance, acid hydrolysis of sucrose requires activation energy of 6.22 kJ/mol, but when hydrolyzed using sucrase enzyme, the activation energy is 2.15 kJ/mol.
Step by step answer:The biochemical reactions occurring within the body is catalyzed by globular proteins called enzymes. They only act by catalyzing the chemical reaction and do not undergo any changes themselves. A particular reaction and substrate have a specific enzyme. The enzymes are named based on the reaction or compound they participate in. Enzymes are often termed as biocatalysts. For instance, the hydrolysis of maltose to glucose is catalyzed by the enzyme maltase.
${{C}_{12}}{{H}_{22}}{{O}_{11}}\xrightarrow{maltase}2{{C}_{6}}{{H}_{12}}{{O}_{6}}$
$Maltose\xrightarrow{maltase}Glu\cos e$
-Ligase: Ligase can be defined as an enzyme that can be utilized to catalyze the fusion of two large molecules by the formation of a new chemical compound. This is usually accompanied by hydrolysis. Ligases such as DNA ligase are enzymes that are utilized in molecular biology laboratories where these enzymes assist in recombinant DNA experiments including DNA replication, DNA repair, and joining DNA fragments. This is achieved by the catalyzed formation of a phosphodiester bond.
-Isomerase: Enzyme that assists in the conversion of a molecule from one isomer to another with the end product having the same molecular formula but different spatial arrangements are called isomerase. Intramolecular rearrangements involve the breaking and formation of bonds. This is facilitated by isomerases. For instance, the glycolysis process involves the conversion of glucose-6-phosphate to fructose-6-phosphate in presence of phosphoglucose isomerase catalyst.

-Oxidoreductase: Transfer of electrons from the reductant to the oxidant is catalyzed by the enzyme oxidoreductase. The reductant is also known as an electron donor, while oxidant is referred to as the electron acceptor. Oxidoreductase enzymes utilize cofactors such as NADP or NAD+.
-Hydrolase: Hydrolase is a biochemical catalyst designed to break a chemical bond through hydrolysis. Hydrolysis is defined as the addition of a water molecule to a chemical process. This results in the breakdown of larger molecules to smaller compounds or molecules.
Therefore the correct option is B, i.e., Isomerase
Note:For the progress of a chemical or biochemical reaction, only a small amount of enzyme is required. Enzymes are believed to decrease the activation energy’s magnitude. For instance, acid hydrolysis of sucrose requires activation energy of 6.22 kJ/mol, but when hydrolyzed using sucrase enzyme, the activation energy is 2.15 kJ/mol.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

Which among the following are examples of coming together class 11 social science CBSE




