Elimination reaction of cis- and trans $-1-$ Bromo $-2-$ methylcyclohexanes with the \[NaOEt\] in \[EtOH\] can give the same or different main product, \[1-\] methylcyclohexene (1) or \[3-\]methylcyclohexene (2). Which of (a)-(d) indicates the main products?
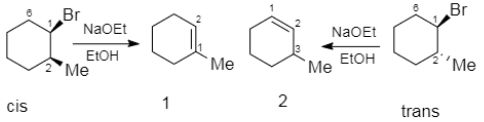
A.$1$ from the cis and $2$ from the trans substrate
B.$2$ from the cis and $1$ from the trans substrate
C.$2$ both from the cis and the trans substrate
D.$1$ both from the cis and the trans substrate

Answer
580.8k+ views
Hint:The chair conformation of the cyclohexane explains the axial and equatorial bonds which are present in the cis- and trans- structures of a substituted cyclohexane.
These confirmation can help us predict the product of an elimination reaction and the position of the double bonds in the products.
Complete step-by-step answer:The two reactants cis and the trans $-1-$ Bromo $-2-$ methylcyclohexanes after individually reacting with sodium ethanoate and ethanol. Now, as we can see there are two reactions which are indicated in the question, in which the reactants differ only in their special arrangements. One of them hence becomes cis and the other becomes trans. In this reaction as we can see that the bromine is the leaving group and the removal of the bromine group would establish the formation of a double bond between the two adjacent carbons.
Unlike structures having an open chain, the cyclic compounds are usually known to have restricted the spatial arrangements of substituents of the ring to relatively lesser orientations. As a consequence of this, reactions which are conducted on these kind of substrates often give us the information about the orientation which is preferred by reactant species in their transition state.
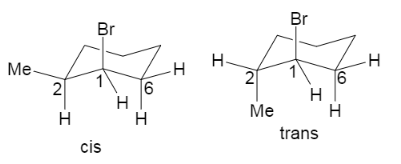
Now, as we can see in the chair conformations the substituents which are present at the positions one and two, takes equatorial and axial position in the cis form, but both of the substituents are in equatorial positions or both are in axial positions in the case of trans form. Hence in the case of cis form, the bromine has the presence of two axial hydrogen which is present in its adjacent carbons $C2$ and $C6$. But in case of the trans structure, there is only one axial hydrogen adjacent to the axial bromine, which is $C6$.
So, the correct option would be A, which says $1$ from the cis and $2$ from the trans substrate.
Note:When a trans cyclohexane undergoes elimination reaction it develops a double bond in the $2-3$ position of the other substituent.
But when a cis cyclohexane undergoes elimination reaction, it develops a double bond in the adjacent position, and this can be understood by the use of chair conformation of the cyclohexane.
These confirmation can help us predict the product of an elimination reaction and the position of the double bonds in the products.
Complete step-by-step answer:The two reactants cis and the trans $-1-$ Bromo $-2-$ methylcyclohexanes after individually reacting with sodium ethanoate and ethanol. Now, as we can see there are two reactions which are indicated in the question, in which the reactants differ only in their special arrangements. One of them hence becomes cis and the other becomes trans. In this reaction as we can see that the bromine is the leaving group and the removal of the bromine group would establish the formation of a double bond between the two adjacent carbons.
Unlike structures having an open chain, the cyclic compounds are usually known to have restricted the spatial arrangements of substituents of the ring to relatively lesser orientations. As a consequence of this, reactions which are conducted on these kind of substrates often give us the information about the orientation which is preferred by reactant species in their transition state.

Now, as we can see in the chair conformations the substituents which are present at the positions one and two, takes equatorial and axial position in the cis form, but both of the substituents are in equatorial positions or both are in axial positions in the case of trans form. Hence in the case of cis form, the bromine has the presence of two axial hydrogen which is present in its adjacent carbons $C2$ and $C6$. But in case of the trans structure, there is only one axial hydrogen adjacent to the axial bromine, which is $C6$.
So, the correct option would be A, which says $1$ from the cis and $2$ from the trans substrate.
Note:When a trans cyclohexane undergoes elimination reaction it develops a double bond in the $2-3$ position of the other substituent.
But when a cis cyclohexane undergoes elimination reaction, it develops a double bond in the adjacent position, and this can be understood by the use of chair conformation of the cyclohexane.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Find the value of the expression given below sin 30circ class 11 maths CBSE

Name the Largest and the Smallest Cell in the Human Body ?

What organs are located on the left side of your body class 11 biology CBSE

What is the weight in lbs of a 75 Kg person class 11 physics CBSE

What is the percentage of photosynthetically active class 11 biology CBSE

How do I convert ms to kmh Give an example class 11 physics CBSE




