How many electrons are present in the M-shell of an element with atomic number 20?
A) 8
B) 6
C) 18
D) 2
Answer
550.6k+ views
Hint: K-shell is the first energy level and can have a maximum of 2 electrons. L-shell is the Second energy level and can have a maximum of 8 electrons. M-shell is the third energy level and can have a maximum of 18 electrons.
Complete step by step answer:
In an atom, the electrons surround the nucleus in different orbitals or energy levels. These orbitals are known as either first, second, third ... energy levels or K,L,M, N... shells. K-shell is the first energy level and can have a maximum of 2 electrons. L-shell is the second energy level and can have a maximum of 8 electrons. M-shell is the third energy level and can have a maximum of 8 electrons.
The atomic number of an element is 20. Atomic number is equal to the number of electrons present in a neutral atom. Thus, the element with atomic number 20 has 20 electrons. Out of 20 electrons, 2 electrons are present in the K-shell and 8 electrons are present in the L-shell. Out of the remaining 20-(2+8)=10 electrons, 8 electrons are present in the M-shell and 2 electrons are present in the N shell.
Hence, 8 electrons are present in the M-shell of an element with atomic number 20.
The structure of atom is as shown below:
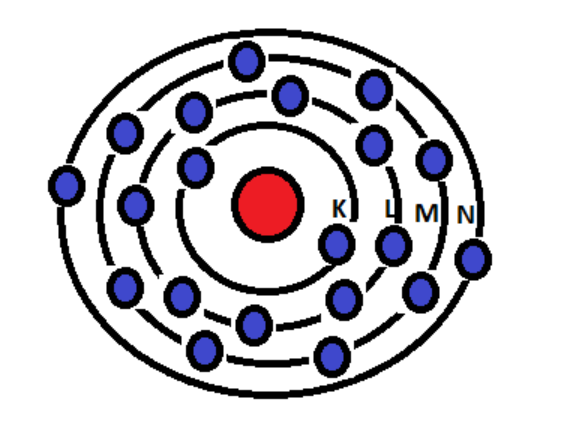
Thus, option A is the correct answer.
Additional information: The electronic configuration of the element with atomic number 20 is \[1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^2}\].
Note:
Do not put more than 2 electrons in K-shell and more than 8 electrons each in L and M-shells.
Complete step by step answer:
In an atom, the electrons surround the nucleus in different orbitals or energy levels. These orbitals are known as either first, second, third ... energy levels or K,L,M, N... shells. K-shell is the first energy level and can have a maximum of 2 electrons. L-shell is the second energy level and can have a maximum of 8 electrons. M-shell is the third energy level and can have a maximum of 8 electrons.
The atomic number of an element is 20. Atomic number is equal to the number of electrons present in a neutral atom. Thus, the element with atomic number 20 has 20 electrons. Out of 20 electrons, 2 electrons are present in the K-shell and 8 electrons are present in the L-shell. Out of the remaining 20-(2+8)=10 electrons, 8 electrons are present in the M-shell and 2 electrons are present in the N shell.
Hence, 8 electrons are present in the M-shell of an element with atomic number 20.
The structure of atom is as shown below:

Thus, option A is the correct answer.
Additional information: The electronic configuration of the element with atomic number 20 is \[1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^2}\].
Note:
Do not put more than 2 electrons in K-shell and more than 8 electrons each in L and M-shells.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

How do I convert ms to kmh Give an example class 11 physics CBSE

Mention the important general characters of Urocho class 11 biology CBSE

Define pneumatic bones class 11 biology CBSE

10 examples of diffusion in everyday life

The relation between molarity M and molality m is given class 11 chemistry CBSE




