Draw the structure of the following
$HCl{O_3}$
Answer
593.1k+ views
Hint:First we have to know what is the central atom in the given compound and then what is the valency of the central atom, what is the hybridisation of the compound, what is the geometry and structure of the compound.
Complete answer:
So, starting from finding the central atom, here the central atom is chlorine, because if we try to make the bonds given, only chlorine will satisfy.
So, now the question is how many valence electrons in the chlorine atom or what is the valency of the chlorine, (because valency is the number of valence electrons present in the outermost valence shell).
Chlorine is having an atomic number of $17$ . Hence, the valency of the chlorine will be $7$ .Now, as chlorine has $7$ valence electrons, it makes $2$ double bonds with oxygen ( $2$ sigma and $2$ pi bonds), one with hydroxide ( $1$ sigma bond) leaving a lone pair on the chlorine atom.
So, the hybridisation of the compound will be $s{p^3}$ . So, the geometry of the compound will be tetrahedral. But due to the presence of a lone pair in the central atom the structure of the compound will be pyramidal as in the place of the one bond there is an lone pair, and we know that in the structure the lone pair is not shown. So, the final structure of the molecule would be pyramidal.
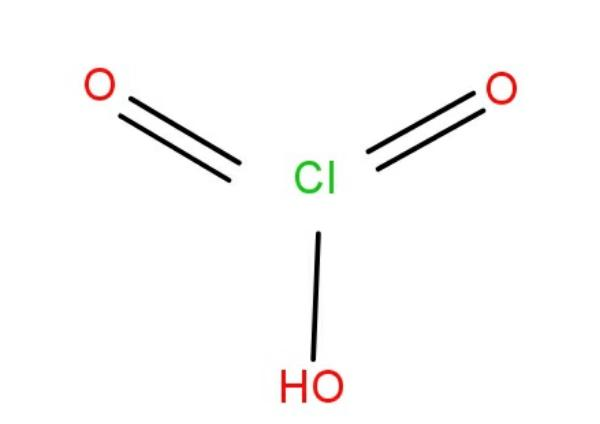
The structure is as shown in figure below:
Note:
When drawing any structure keep the points in mind that the valency of the central atom is complete and presence of lone pair take part while writing geometry, but while drawing the structure it would not be shown. Due to the presence of lone pairs, the geometry of the compound will be distorted.
Complete answer:
So, starting from finding the central atom, here the central atom is chlorine, because if we try to make the bonds given, only chlorine will satisfy.
So, now the question is how many valence electrons in the chlorine atom or what is the valency of the chlorine, (because valency is the number of valence electrons present in the outermost valence shell).
Chlorine is having an atomic number of $17$ . Hence, the valency of the chlorine will be $7$ .Now, as chlorine has $7$ valence electrons, it makes $2$ double bonds with oxygen ( $2$ sigma and $2$ pi bonds), one with hydroxide ( $1$ sigma bond) leaving a lone pair on the chlorine atom.
So, the hybridisation of the compound will be $s{p^3}$ . So, the geometry of the compound will be tetrahedral. But due to the presence of a lone pair in the central atom the structure of the compound will be pyramidal as in the place of the one bond there is an lone pair, and we know that in the structure the lone pair is not shown. So, the final structure of the molecule would be pyramidal.
The structure is as shown in figure below:

Note:
When drawing any structure keep the points in mind that the valency of the central atom is complete and presence of lone pair take part while writing geometry, but while drawing the structure it would not be shown. Due to the presence of lone pairs, the geometry of the compound will be distorted.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE




