Draw the structure of the compound:
${\text{4 - tert - butyl - 3 - iodoheptane}}$
Answer
573k+ views
Hint:In organic chemistry, we have three different ways to draw organic molecules including structural formula, condensed formula, and skeletal structures (also known as line – bond structures or line formulas). ${\text{4 - tert - butyl - 3 - iodoheptane}}$ is an organic halogen compound, to draw the structure format with root, prefix, and suffix.
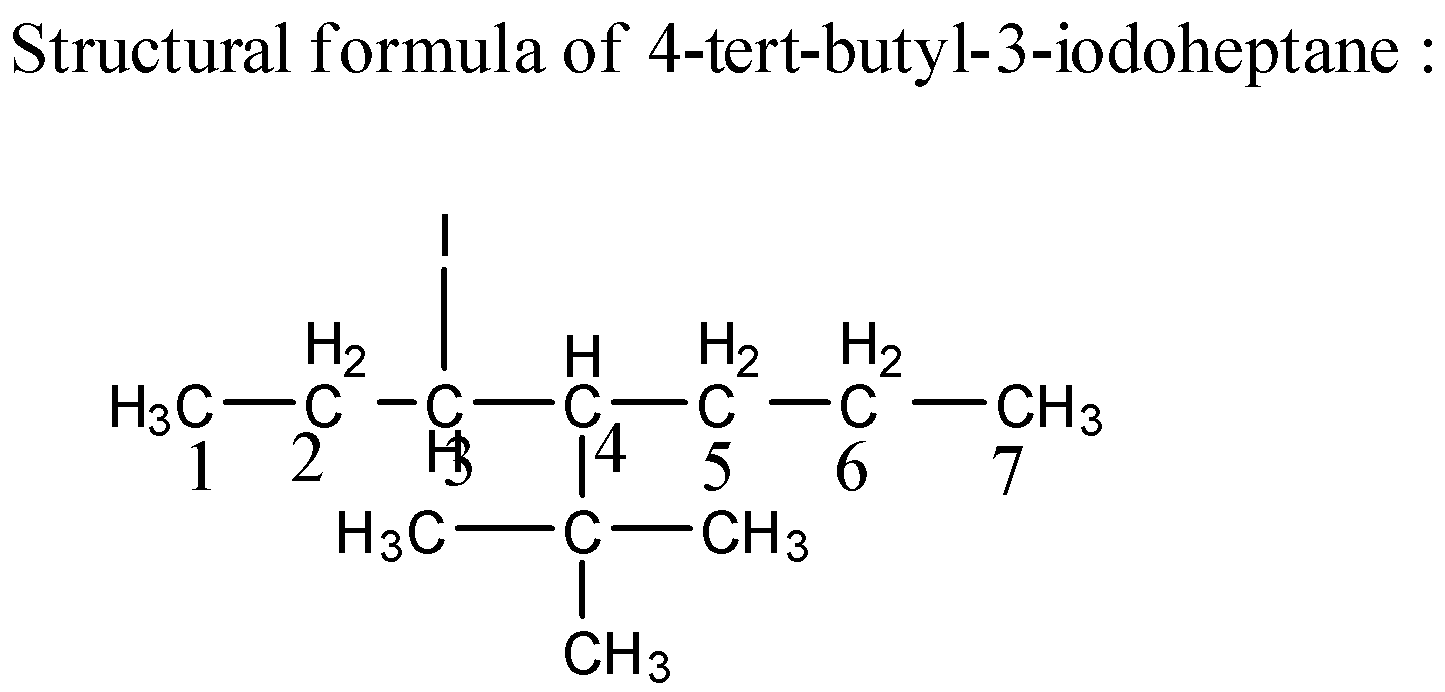
Complete step by step answer:1) First of all, in the given compound the end word is -hept, which shows the longest carbon chain must have seven carbon atoms. There are two substituent presents in a structure, one at carbon atom number ${\text{3}}$ and one at ${\text{4}}$. The carbon numbered ${\text{3}}$ contains the substituent Iodine and the carbon number ${\text{4}}$ contains the substituent tert butyl.
2) The IUPAC name starts from the number of carbon atoms bearing the substituent followed by the name of the substituent. The substituents that are attached to the longest chain comes first as per alphabetical order and it is named first. Then the word root hept comes followed by the suffix -ane which means all the bonds are single bonds.
3) Structural formula- displays the atoms of the molecule in the order they are bonded.
Structural Formula of ${\text{4 - tert - butyl - 3 - iodoheptane}}$ is as below,
4) The condensed formula displays the order of atoms similar to the structural formula but is written in a single line and makes it easier and faster to write out. The condensed Formula of ${\text{4 - tert - butyl - 3 - iodoheptane}}$ is $C{H_3} - C{H_2} - CH(I) - C(CC{H_3}) - C{H_2} - C{H_2} - C{H_3}$
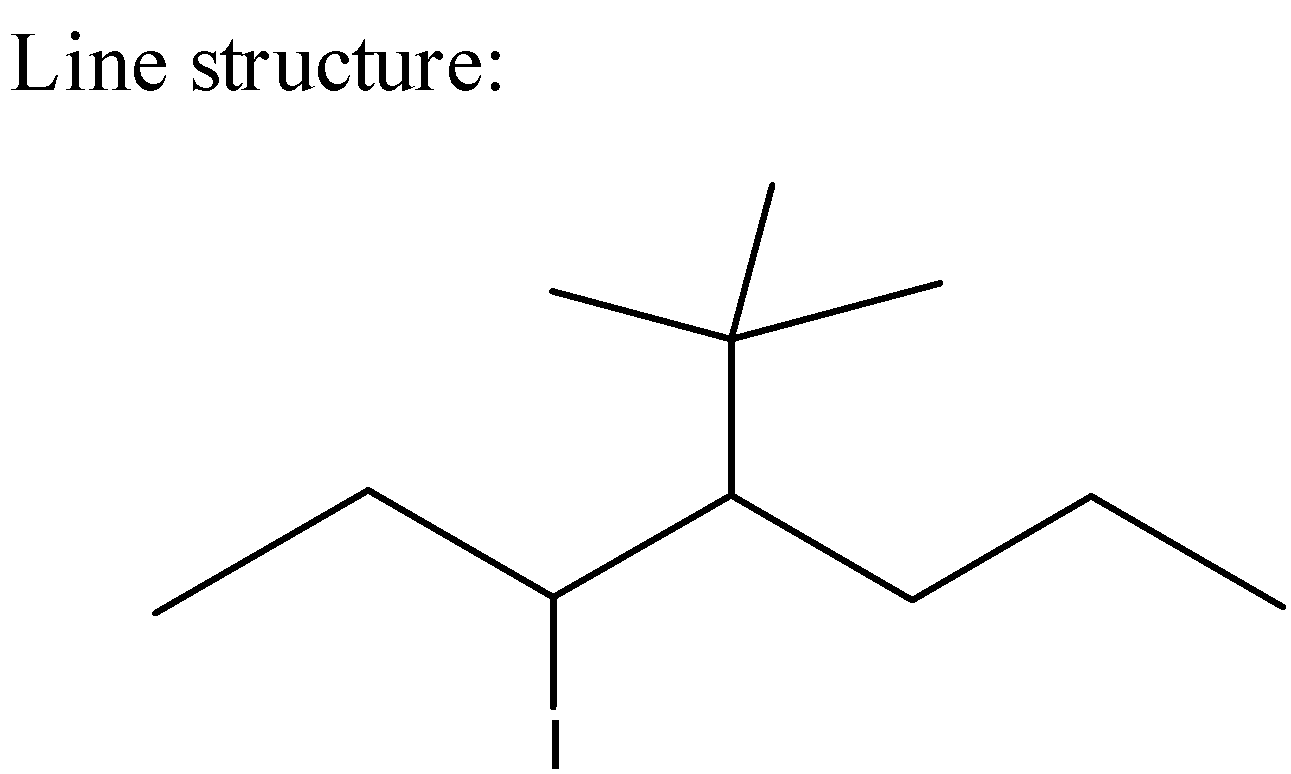
5) The line formulas are used to write carbon and hydrogen atoms more significantly by replacing the letter "C" with lines.
Line Formula is as below,
Note:
The numbering given in an IUPAC name shows the atoms or groups that are attached to the carbon atoms in the longest chain. It is very important to write a structure based on the IUPAC name as IUPAC naming gives the identity to a chemical structure.
Complete step by step answer:1) First of all, in the given compound the end word is -hept, which shows the longest carbon chain must have seven carbon atoms. There are two substituent presents in a structure, one at carbon atom number ${\text{3}}$ and one at ${\text{4}}$. The carbon numbered ${\text{3}}$ contains the substituent Iodine and the carbon number ${\text{4}}$ contains the substituent tert butyl.
2) The IUPAC name starts from the number of carbon atoms bearing the substituent followed by the name of the substituent. The substituents that are attached to the longest chain comes first as per alphabetical order and it is named first. Then the word root hept comes followed by the suffix -ane which means all the bonds are single bonds.
3) Structural formula- displays the atoms of the molecule in the order they are bonded.
Structural Formula of ${\text{4 - tert - butyl - 3 - iodoheptane}}$ is as below,

4) The condensed formula displays the order of atoms similar to the structural formula but is written in a single line and makes it easier and faster to write out. The condensed Formula of ${\text{4 - tert - butyl - 3 - iodoheptane}}$ is $C{H_3} - C{H_2} - CH(I) - C(CC{H_3}) - C{H_2} - C{H_2} - C{H_3}$
5) The line formulas are used to write carbon and hydrogen atoms more significantly by replacing the letter "C" with lines.
Line Formula is as below,

Note:
The numbering given in an IUPAC name shows the atoms or groups that are attached to the carbon atoms in the longest chain. It is very important to write a structure based on the IUPAC name as IUPAC naming gives the identity to a chemical structure.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




