Draw the structure diagram of ammonia molecule as per the valence-shell electron pair repulsion theory.
Answer
625.2k+ views
Hint: Valence shell electron pair repulsion theory, or VSEPR theory, is a concept used in chemistry to determine the structure of single molecules from the number of pairs of electrons surrounding their central atoms. The formula of an ammonia molecule is $N{{H}_{3}}$.
Complete answer:
Valence-shell electron pair repulsion theory: The Valence Shell Electron Pair Repulsion Theory abbreviated as VSEPR theory is based on the premise that there is a repulsion between the pairs of valence electrons in all atoms, and the atoms will always tend to arrange themselves in a manner in which this electron pair repulsion is minimalized. This arrangement of the atom determines the geometry of the resulting molecule.
In a molecule two types of electron pair are there, bond pair and lone pair.
Bond pair: it is under the influence of two nuclei.
Loan pair: It is under the influence of one nucleus.
The repulsion between electron pairs follows the order, lone pair – lone pair > lone pair – bond pair > bond pair – bond pair, because different types of repulsion distortion in geometry will be observed from the standard geometry w.r.t hybridization.
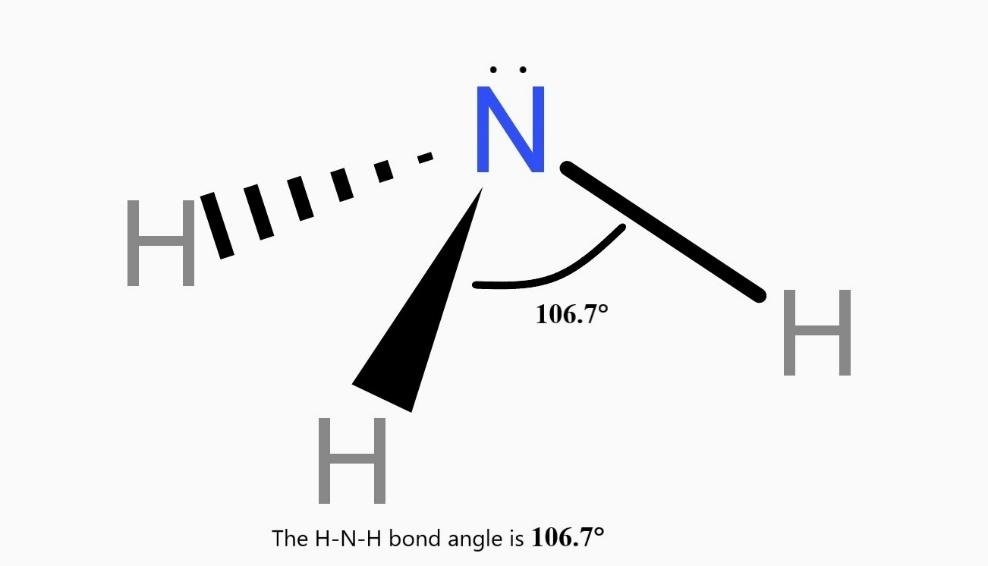
Ammonia is made up of 1 atom of nitrogen and 3 atoms of hydrogen. Nitrogen has 8 electrons around it i.e. it has 4 electron pairs. These pairs are arranged tetrahedrally. Of the 4 pairs: 1 pair is a lone pair. So, the resulting shape is a trigonal pyramid.
Note: The VSEPR theory assumes that each atom in a molecule will achieve a geometry that minimizes the repulsion between electrons in the valence shell of that atom.
The structure of ammonia molecules as per the valence-shell electron pair repulsion theory is trigonal pyramid.
The H-N-H bond angle is 106.7°.
Complete answer:
Valence-shell electron pair repulsion theory: The Valence Shell Electron Pair Repulsion Theory abbreviated as VSEPR theory is based on the premise that there is a repulsion between the pairs of valence electrons in all atoms, and the atoms will always tend to arrange themselves in a manner in which this electron pair repulsion is minimalized. This arrangement of the atom determines the geometry of the resulting molecule.
In a molecule two types of electron pair are there, bond pair and lone pair.
Bond pair: it is under the influence of two nuclei.
Loan pair: It is under the influence of one nucleus.
The repulsion between electron pairs follows the order, lone pair – lone pair > lone pair – bond pair > bond pair – bond pair, because different types of repulsion distortion in geometry will be observed from the standard geometry w.r.t hybridization.
Ammonia is made up of 1 atom of nitrogen and 3 atoms of hydrogen. Nitrogen has 8 electrons around it i.e. it has 4 electron pairs. These pairs are arranged tetrahedrally. Of the 4 pairs: 1 pair is a lone pair. So, the resulting shape is a trigonal pyramid.

Note: The VSEPR theory assumes that each atom in a molecule will achieve a geometry that minimizes the repulsion between electrons in the valence shell of that atom.
The structure of ammonia molecules as per the valence-shell electron pair repulsion theory is trigonal pyramid.
The H-N-H bond angle is 106.7°.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




