Draw the electron dot structure of ethane.
Answer
598.5k+ views
Hint: For the required structure, we need to consider the Lewis structure. And for that we have to consider the number of the valence electrons for the given molecule and draw the structure. One needs to know that; an ethane molecule has two carbon atoms and six hydrogen atoms. The homologous series formula for alkanes is to be followed here to figure out the chemical formula of the given molecule. As ethane is an alkane, it will follow the alkane formula ${{C}_{n}}{{H}_{2n+2}}$.
Complete Solution :
We have to draw the dot structure of ethane.
The chemical formula of ethane is ${{C}_{2}}{{H}_{6}}$ and its chemical structure for this molecule is given below:
$C{{H}_{3}}-C{{H}_{3}}$
All the bonds present in ethane molecules are covalent bonds. The electron dot structure for the ethane molecule is:
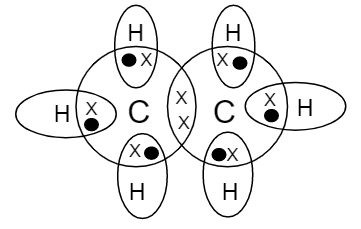
- We should know that, on the basis of outermost shell electron configuration of carbon, there are four valence electrons for carbon whereas there is only one valence electron for hydrogen atom. These electrons are shared as shown in the diagram through a covalent bond. Each carbon atom is surrounded by three hydrogen atoms as shown.
Note: Some interesting facts about ethane are it is the second in the list of homologous series of alkane. The chemical formula for the homologous series is ${{C}_{n}}{{H}_{2n+2}}$. First one being, methane whose chemical formula is $C{{H}_{4}}$, the other one in this series are propane (${{C}_{3}}{{H}_{8}}$), butane (${{C}_{4}}{{H}_{10}}$) and so on.
Complete Solution :
We have to draw the dot structure of ethane.
The chemical formula of ethane is ${{C}_{2}}{{H}_{6}}$ and its chemical structure for this molecule is given below:
$C{{H}_{3}}-C{{H}_{3}}$
All the bonds present in ethane molecules are covalent bonds. The electron dot structure for the ethane molecule is:

- We should know that, on the basis of outermost shell electron configuration of carbon, there are four valence electrons for carbon whereas there is only one valence electron for hydrogen atom. These electrons are shared as shown in the diagram through a covalent bond. Each carbon atom is surrounded by three hydrogen atoms as shown.
Note: Some interesting facts about ethane are it is the second in the list of homologous series of alkane. The chemical formula for the homologous series is ${{C}_{n}}{{H}_{2n+2}}$. First one being, methane whose chemical formula is $C{{H}_{4}}$, the other one in this series are propane (${{C}_{3}}{{H}_{8}}$), butane (${{C}_{4}}{{H}_{10}}$) and so on.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

The number of chromosomes in male grasshopper is a class 12 biology CBSE




