Draw the electron dot structure of Ethane.
Answer
584.1k+ views
Hint In order to draw the structure, consider the number of valence electrons for the given molecule and then draw the structure. We should know that an ethane molecule has 6 hydrogen atoms and 2 carbon atoms. The formula of ethane is found to be ${{C}_{n}}{{H}_{2n+2}}$, that is we can say that the homologous series of formula of alkanes is followed here because ethane is an alkane.
Complete Step by step solution:
- Let us first draw the electron dot structure of Ethane and then will discuss in brief about its structure:
- We can write the chemical structure for ethane molecule as:
$C{{H}_{3}}-C{{H}_{3}}$
- As we know that all the bonds that are present in ethane molecules are covalent bonds. We can draw the electron dot structure of ethane molecule as:
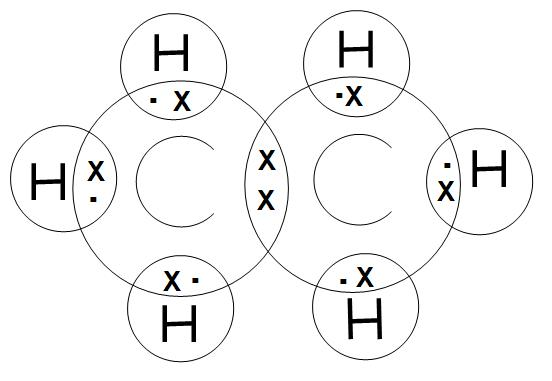
- It is found that on the basis of outermost valence shell electronic configuration of carbon, there are four valence electrons. Whereas, there is only 1 valence electron for hydrogen. We can see in the above structure that they are shared by a covalent bond.
- And we can see that each carbon atom is basically surrounded by three hydrogen atoms.
Note:
- As we know that there are some interesting facts found about ethane that it is the second in the homologous series of alkane. The first one found to be methane, which is further followed by the other one in the series are propane, butane and so on.
Complete Step by step solution:
- Let us first draw the electron dot structure of Ethane and then will discuss in brief about its structure:
- We can write the chemical structure for ethane molecule as:
$C{{H}_{3}}-C{{H}_{3}}$
- As we know that all the bonds that are present in ethane molecules are covalent bonds. We can draw the electron dot structure of ethane molecule as:

- It is found that on the basis of outermost valence shell electronic configuration of carbon, there are four valence electrons. Whereas, there is only 1 valence electron for hydrogen. We can see in the above structure that they are shared by a covalent bond.
- And we can see that each carbon atom is basically surrounded by three hydrogen atoms.
Note:
- As we know that there are some interesting facts found about ethane that it is the second in the homologous series of alkane. The first one found to be methane, which is further followed by the other one in the series are propane, butane and so on.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




