Draw the electron dot structure of ethane molecule (${{C}_{2}}{{H}_{6}}$ ).
Answer
555.1k+ views
Hint: For the required structure, we need to consider the Lewis structure. For that consider the number of valence electrons for the given molecule and draw the structure. Also, one needs to know that an ethane molecule has 2 carbon atoms and 6 hydrogen atoms. The homologous series formula for alkanes is followed here as Ethane is an alkane. The formula is ${{C}_{n}}{{H}_{2n+2}}$
Complete Step by Step Solution:
The chemical structure for the ethane molecule is given below:
$C{{H}_{3}}-C{{H}_{3}}$
All the bonds present in the ethane molecule are covalent bonds. The Electron dot structure for ethane molecule is:
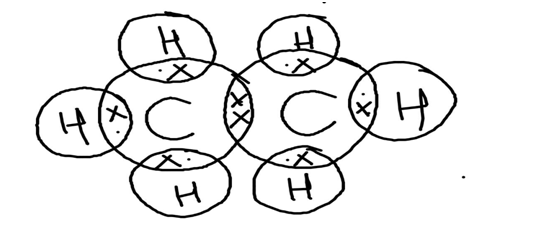
● We know that on the basis of outermost shell electron configuration of Carbon, there is 4 valence electrons for Carbon whereas there is only 1 valence electron for Hydrogen. They are shared as shown in the structure through a covalent bond.
● Each carbon atom is surrounded by three hydrogen atoms as shown.
Note: Some interesting facts about Ethane are that it is the second in the list of Homologous series of Alkane. First one being, Methane whose chemical formula is $C{{H}_{4}}$ , the other ones in this series are Propane (${{C}_{3}}{{H}_{8}}$ ), Butane (${{C}_{4}}{{H}_{10}}$ ) and so on.
Complete Step by Step Solution:
The chemical structure for the ethane molecule is given below:
$C{{H}_{3}}-C{{H}_{3}}$
All the bonds present in the ethane molecule are covalent bonds. The Electron dot structure for ethane molecule is:

● We know that on the basis of outermost shell electron configuration of Carbon, there is 4 valence electrons for Carbon whereas there is only 1 valence electron for Hydrogen. They are shared as shown in the structure through a covalent bond.
● Each carbon atom is surrounded by three hydrogen atoms as shown.
Note: Some interesting facts about Ethane are that it is the second in the list of Homologous series of Alkane. First one being, Methane whose chemical formula is $C{{H}_{4}}$ , the other ones in this series are Propane (${{C}_{3}}{{H}_{8}}$ ), Butane (${{C}_{4}}{{H}_{10}}$ ) and so on.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources

The speaker of the Lok Sabha is elected by the APresident class 10 social science CBSE

Complete the sentence with the most appropriate word class 10 english CBSE




