Double bond equivalent (degree of unsaturation) of (A) is-
(A) 1
(B) 2
(C) 3
(D) 4

Answer
629.7k+ views
Hint: A measurement which determines the total number of rings and α bonds is the degree of unsaturation. A formula for drawing chemical structures is used in organic chemistry. Although it gives no details about those elements separately- the specific numbers of rings or of double bonds (one pi − bond), or of triple bonds (two pi- bonds each) is given individually.
Complete step by step solution:
Formula used: $C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2}$.
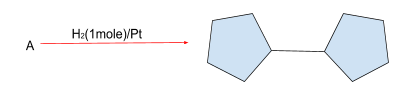
Catalytic hydrogenation of alkenes is caused by ${H_2}/Pt$. Because 1 mole of the reagent is available, only one double bond will be applied to ${H_2}$. So, two cyclopentane rings with a double relation should be present in compound A.
The double bond equivalent in A is three because of the presence of two rings and one dual bond. We may also use the formula to measure it.
The formula used to calculate double bond equivalent is-
$ \Rightarrow C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2}$
Where, C stands for carbon atoms, H for hydrogen atoms, X is for halogens.
Here, the value of C is 10
The value of H is two less than the number of H in the product = 18-2 = 16
Thus, Double bond equivalent is-
$
\Rightarrow C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2} \\
\\
\Rightarrow 10 + 1 - \dfrac{{16}}{2} \\
\\
\Rightarrow 10 + 1 - 8 \\
\\
\Rightarrow 3 \\
$
Hence, it is clear that option C is the correct option.
Note: The number of unsaturation in an organic molecule is DBE or double bond equivalent which is also known as the degree of unsaturation. Unsaturation refers to a double bond or a ring system. For example , 3 double bonds are possible for benzene and 1 ring gives us 4 DBE. A triple bond may also be considered as DBE = 2. X is the total number of halogens in the structure, that is, Cl, Br, F, and I. DBE measurement is not affected by the existence of the oxygen atom.
Complete step by step solution:
Formula used: $C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2}$.
Catalytic hydrogenation of alkenes is caused by ${H_2}/Pt$. Because 1 mole of the reagent is available, only one double bond will be applied to ${H_2}$. So, two cyclopentane rings with a double relation should be present in compound A.
The double bond equivalent in A is three because of the presence of two rings and one dual bond. We may also use the formula to measure it.
The formula used to calculate double bond equivalent is-
$ \Rightarrow C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2}$
Where, C stands for carbon atoms, H for hydrogen atoms, X is for halogens.
Here, the value of C is 10
The value of H is two less than the number of H in the product = 18-2 = 16
Thus, Double bond equivalent is-
$
\Rightarrow C + 1 - \dfrac{H}{2} - \dfrac{X}{2} + \dfrac{N}{2} \\
\\
\Rightarrow 10 + 1 - \dfrac{{16}}{2} \\
\\
\Rightarrow 10 + 1 - 8 \\
\\
\Rightarrow 3 \\
$
Hence, it is clear that option C is the correct option.
Note: The number of unsaturation in an organic molecule is DBE or double bond equivalent which is also known as the degree of unsaturation. Unsaturation refers to a double bond or a ring system. For example , 3 double bonds are possible for benzene and 1 ring gives us 4 DBE. A triple bond may also be considered as DBE = 2. X is the total number of halogens in the structure, that is, Cl, Br, F, and I. DBE measurement is not affected by the existence of the oxygen atom.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE

State the laws of reflection of light

Mention the important general characters of Urocho class 11 biology CBSE

Define pneumatic bones class 11 biology CBSE

Arrange the bond angle order of NO2NO2 NO2 + and NO3 class 11 chemistry CBSE

The relation between molarity M and molality m is given class 11 chemistry CBSE




