Describe the nitrogen cycle with the help of a diagram.
Answer
627.9k+ views
Hint: This cycle is a biogeochemical process through which nitrogen is converted into many forms, consecutively passing from the atmosphere to the soil to the organism and back to the atmosphere.
Complete answer:
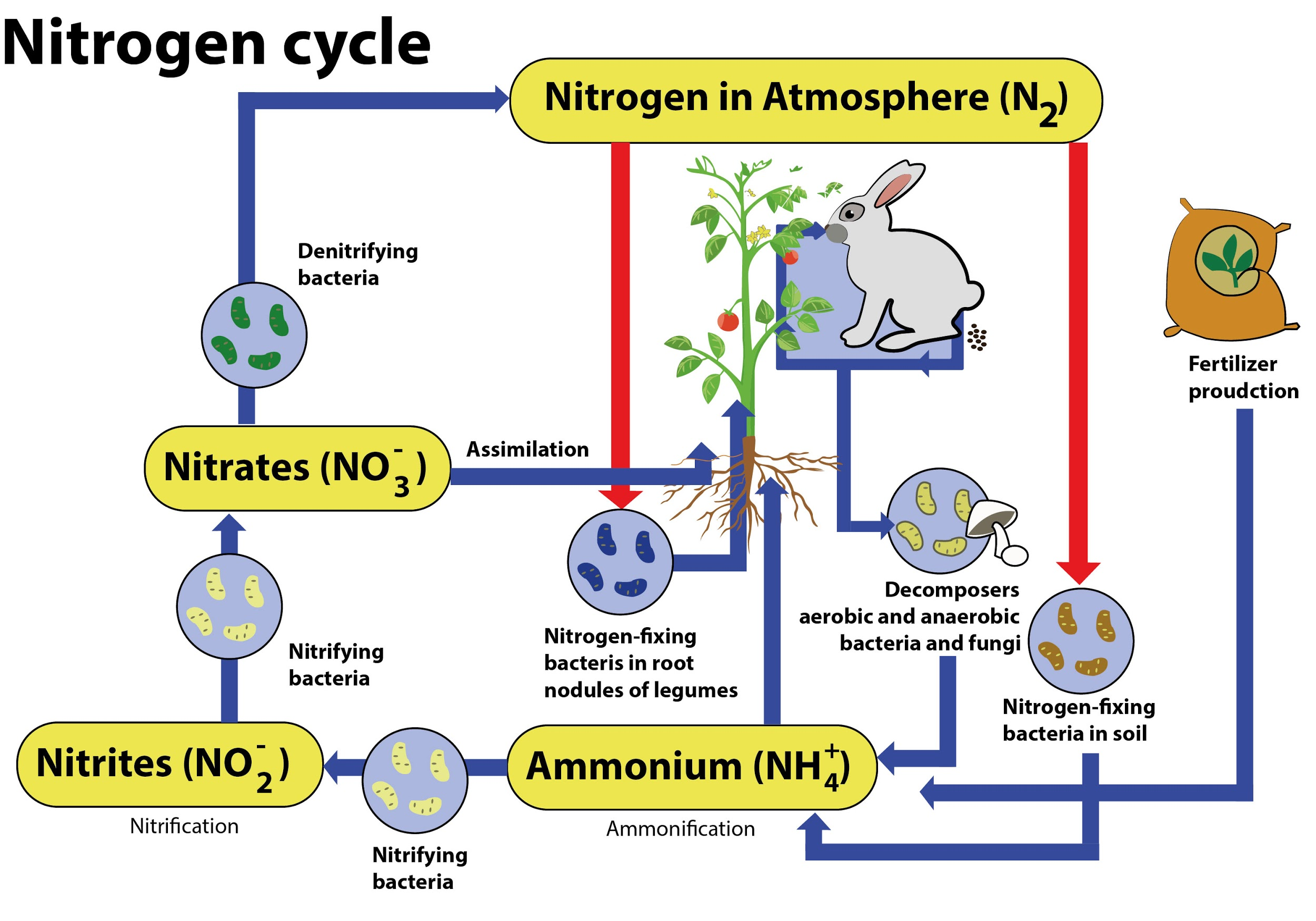
Nitrogen undergoes various sorts of transformation to take care of a balance within the ecosystem. Furthermore, this process extends to varied biomes, with the marine organic process being one of the foremost complicated biogeochemical cycles.
Stages of Nitrogen Cycle
The organic process consists of the subsequent steps – organic process, Nitrification, Assimilation, Ammonification, and Denitrification. These processes take place in several stages and are explained below:
-Nitrogen fixation: It is the initial step of the organic process. Here, Atmospheric nitrogen ($N_{2}$) is converted into the usable form - ammonia ($NH_{3}$).
The entire organic process is completed by symbiotic bacteria which are referred to as Diazotrophs. Azotobacter and Rhizobium also have a major role in this process. These bacteria contain a nitrogenase enzyme that has the potential to mix gaseous nitrogen with hydrogen to make ammonia.
-Nitrification: In this process, the ammonia is converted into nitrate by the presence of bacteria within the soil. By the oxidation of Ammonia with the help of Nitrosomonas bacteria species, nitrites are formed. Later, the produced nitrites are converted into nitrates by Nitrobacter. This conversion is extremely important as ammonia gas is toxic for plants.
The reaction involved in the process of Nitrification is as follows:
$2NH_{4}^{+}+3O_{2}\rightarrow 2NO_{2}^{-}+4H^{+}2H_{2}O$
$2NO_{2}^{-}+O_{2}\rightarrow 2NO_{3}^{-}$
-Assimilation: Primary producers – plants absorb the nitrogen compounds from the soil with the assistance of their roots, which are available within the sort of ammonia, nitrite ions, nitrate ions, or ammonium ions and are used in the formation of the plant and animal proteins. This way, it enters the food web when the primary consumers eat the plants.
-Ammonification: When plants or animals die, the nitrogen present within the organic matter is released back to the soil. The decomposers, namely bacteria or fungi present within the soil, convert the organic matter back to ammonium. This process of decomposition produces ammonia, which is further used for other biological processes.
-Denitrification: Denitrification is the process in which the nitrogen compound makes its way back into the atmosphere by converting nitrate ($NO_{3}^{-}$) into gaseous nitrogen (N). This process of the nitrogen cycle is the final stage and occurs in the absence of oxygen. Denitrification is carried out by the denitrifying bacterial species- Clostridium and Pseudomonas, which will process nitrate to gain oxygen and gives out free nitrogen gas as a byproduct.
Note: -Ammonium is thought to be the preferred source of fixed nitrogen for phytoplankton because its assimilation does not involve a redox reaction and therefore requires little energy
-Between ammonium, nitrite, and nitrate, nitrite has the fastest turnover rate. It can be produced during nitrate assimilation, nitrification, and denitrification; however, it is immediately consumed again.
-The nitrogen gas exists in both organic and inorganic forms. Organic nitrogen exists in living organisms, and they get passed through the food chain by the consumption of other living organisms.
-Inorganic forms of nitrogen are found in abundance in the atmosphere. This nitrogen is made available to plants by symbiotic bacteria which can convert the inert nitrogen into a usable form – such as nitrites and nitrates.
Complete answer:

Nitrogen undergoes various sorts of transformation to take care of a balance within the ecosystem. Furthermore, this process extends to varied biomes, with the marine organic process being one of the foremost complicated biogeochemical cycles.
Stages of Nitrogen Cycle
The organic process consists of the subsequent steps – organic process, Nitrification, Assimilation, Ammonification, and Denitrification. These processes take place in several stages and are explained below:
-Nitrogen fixation: It is the initial step of the organic process. Here, Atmospheric nitrogen ($N_{2}$) is converted into the usable form - ammonia ($NH_{3}$).
The entire organic process is completed by symbiotic bacteria which are referred to as Diazotrophs. Azotobacter and Rhizobium also have a major role in this process. These bacteria contain a nitrogenase enzyme that has the potential to mix gaseous nitrogen with hydrogen to make ammonia.
-Nitrification: In this process, the ammonia is converted into nitrate by the presence of bacteria within the soil. By the oxidation of Ammonia with the help of Nitrosomonas bacteria species, nitrites are formed. Later, the produced nitrites are converted into nitrates by Nitrobacter. This conversion is extremely important as ammonia gas is toxic for plants.
The reaction involved in the process of Nitrification is as follows:
$2NH_{4}^{+}+3O_{2}\rightarrow 2NO_{2}^{-}+4H^{+}2H_{2}O$
$2NO_{2}^{-}+O_{2}\rightarrow 2NO_{3}^{-}$
-Assimilation: Primary producers – plants absorb the nitrogen compounds from the soil with the assistance of their roots, which are available within the sort of ammonia, nitrite ions, nitrate ions, or ammonium ions and are used in the formation of the plant and animal proteins. This way, it enters the food web when the primary consumers eat the plants.
-Ammonification: When plants or animals die, the nitrogen present within the organic matter is released back to the soil. The decomposers, namely bacteria or fungi present within the soil, convert the organic matter back to ammonium. This process of decomposition produces ammonia, which is further used for other biological processes.
-Denitrification: Denitrification is the process in which the nitrogen compound makes its way back into the atmosphere by converting nitrate ($NO_{3}^{-}$) into gaseous nitrogen (N). This process of the nitrogen cycle is the final stage and occurs in the absence of oxygen. Denitrification is carried out by the denitrifying bacterial species- Clostridium and Pseudomonas, which will process nitrate to gain oxygen and gives out free nitrogen gas as a byproduct.
Note: -Ammonium is thought to be the preferred source of fixed nitrogen for phytoplankton because its assimilation does not involve a redox reaction and therefore requires little energy
-Between ammonium, nitrite, and nitrate, nitrite has the fastest turnover rate. It can be produced during nitrate assimilation, nitrification, and denitrification; however, it is immediately consumed again.
-The nitrogen gas exists in both organic and inorganic forms. Organic nitrogen exists in living organisms, and they get passed through the food chain by the consumption of other living organisms.
-Inorganic forms of nitrogen are found in abundance in the atmosphere. This nitrogen is made available to plants by symbiotic bacteria which can convert the inert nitrogen into a usable form – such as nitrites and nitrates.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




