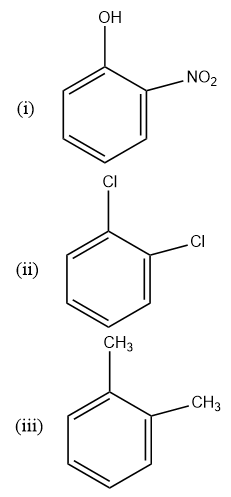
Correct order of dipole moment is:
A. \[i = ii = iii\]
B. \[i < ii < iii\]
C. \[i > ii > iii\]
D. \[ii < iii < i\]

Answer
564k+ views
Hint: Dipole moment is defined as the separation of the charges in the system. Both of the ionic and the covalently bonded compound tends to have the dipole moment. The reason for the cause of the dipole moment is the difference between the electronegativity of the chemically bonded atoms present in the molecule.
Complete step by step answer:
So the name of the compound (I)is the o- nitrophenol. The name of the (II)is 1,2 dichlorobenzene and the (III)is the 1,2 dimethylbenzene. The dipole moment as I said depends upon the electronegativity of the atom. So the electronegativity of the oxygen and nitrogen is greater than that of the chlorine and the methyl group. The dipole moment is pointed from the less electronegative atom towards the more electronegative atom. Due to the presence of the –R effect in the nitro group and the presence of the +R effect of the phenol the compound \[(i)\] will have more dipole moment and then the 1,2 dichlorobenzene will have lees dipole than the \[(i)\] but more than \[(iii)\] as it is more electronegative whereas \[(iii)\] will have the least dipole moment as it has the methyl group which is the least electronegative and is the electron donating group.
So the order is \[i > ii > iii\] So the correct option is option c.
Note: The bond dipole moment tends to differ from that of the total dipole moment in the polyatomic molecule. The bond dipole moment is defined as the dipole moment which is present between the single bonded diatomic molecules. Whereas the total dipole moment in the polyatomic molecule is defined as the vector sum of all the bond dipoles present in the molecule.
Complete step by step answer:
So the name of the compound (I)is the o- nitrophenol. The name of the (II)is 1,2 dichlorobenzene and the (III)is the 1,2 dimethylbenzene. The dipole moment as I said depends upon the electronegativity of the atom. So the electronegativity of the oxygen and nitrogen is greater than that of the chlorine and the methyl group. The dipole moment is pointed from the less electronegative atom towards the more electronegative atom. Due to the presence of the –R effect in the nitro group and the presence of the +R effect of the phenol the compound \[(i)\] will have more dipole moment and then the 1,2 dichlorobenzene will have lees dipole than the \[(i)\] but more than \[(iii)\] as it is more electronegative whereas \[(iii)\] will have the least dipole moment as it has the methyl group which is the least electronegative and is the electron donating group.
So the order is \[i > ii > iii\] So the correct option is option c.
Note: The bond dipole moment tends to differ from that of the total dipole moment in the polyatomic molecule. The bond dipole moment is defined as the dipole moment which is present between the single bonded diatomic molecules. Whereas the total dipole moment in the polyatomic molecule is defined as the vector sum of all the bond dipoles present in the molecule.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




