What is the correct formula of dinitrogen sulfide?
A) \[N{S_2}\]
B) \[N{S_{}}\]
C) \[{N_2}S\]
D) None of these
Answer
549k+ views
Hint: We have to remember that it is easy to identify the chemical formula by looking at the chemical name. For example: \[N\]can be written as nitrogen whereas \[{O_2}\] is Dioxide, So the formula for \[N{O_2}\] is nitrogen dioxide. Molecular weight of \[{N_2}S\] is $60g/mol$.
Complete answer:
It is easy to find the molecular formula, since it is dinitrogen then there must be two nitrogen atoms present in a molecule.
Option A) this is an incorrect option as \[N{S_2}\] represents disulfide instead of dinitrogen thus we can easily neglect this option.
Option B) This is an incorrect option as \[N{S_{}}\]does not represent dinitrogen sulfide as it does not have \[{N_2}\] in its formula.
Option C) this is a correct option as \[{N_2}S\] represents dinitrogen sulfide.
Dinitrogen sulfide has a structural formula
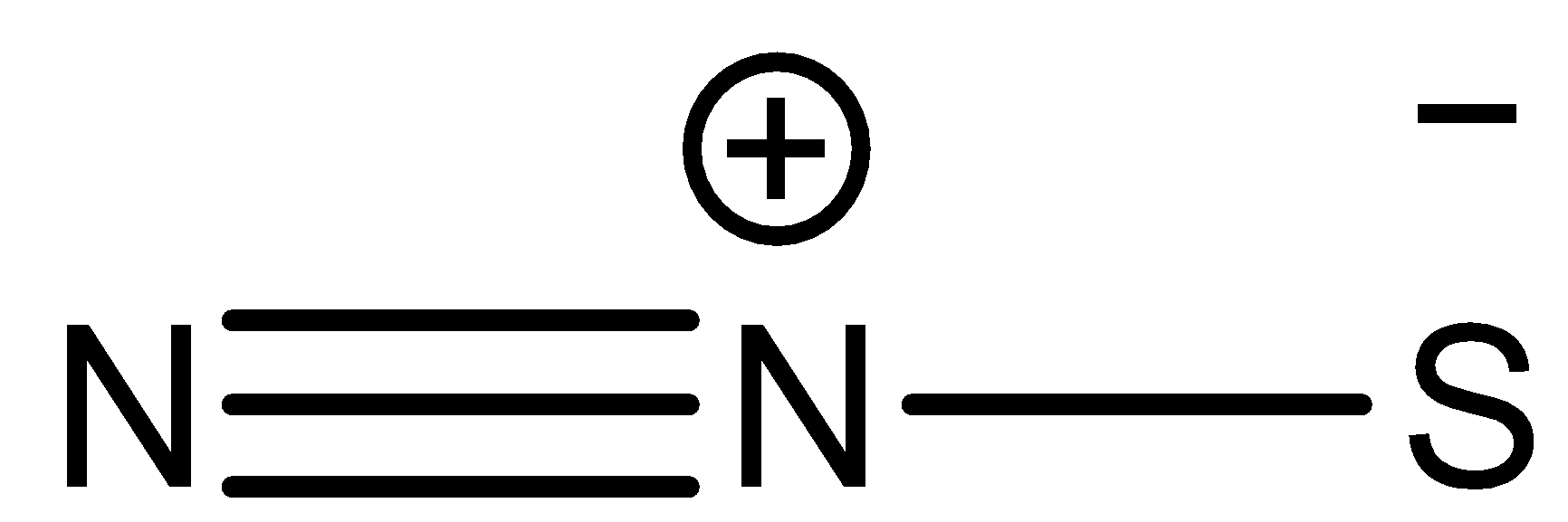
\[{N_2}S\] has a molecular weight$ = 60g/mol$. It is a highly polar molecule due to the presence of formal charges on it. It is a linear molecule which is considered to be stable and unreactive at room temperature.
Option D) this is an incorrect option as we got option C as a correct option.
Note:
We have to remember that the dinitrogen sulfide molecule is highly polar in nature since it has two opposite charges to attract between the bonds that hold an atom or a molecule. This molecule is a linear molecule that can be considered to be stable and unreactive at room temperature. In this molecule since sulphur is less electronegative than nitrogen thus electrons will prefer to lie on nitrogen atoms than sulphur.
Complete answer:
It is easy to find the molecular formula, since it is dinitrogen then there must be two nitrogen atoms present in a molecule.
Option A) this is an incorrect option as \[N{S_2}\] represents disulfide instead of dinitrogen thus we can easily neglect this option.
Option B) This is an incorrect option as \[N{S_{}}\]does not represent dinitrogen sulfide as it does not have \[{N_2}\] in its formula.
Option C) this is a correct option as \[{N_2}S\] represents dinitrogen sulfide.
Dinitrogen sulfide has a structural formula

\[{N_2}S\] has a molecular weight$ = 60g/mol$. It is a highly polar molecule due to the presence of formal charges on it. It is a linear molecule which is considered to be stable and unreactive at room temperature.
Option D) this is an incorrect option as we got option C as a correct option.
Note:
We have to remember that the dinitrogen sulfide molecule is highly polar in nature since it has two opposite charges to attract between the bonds that hold an atom or a molecule. This molecule is a linear molecule that can be considered to be stable and unreactive at room temperature. In this molecule since sulphur is less electronegative than nitrogen thus electrons will prefer to lie on nitrogen atoms than sulphur.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




