How will you convert Benzene to Benzoic acid ?
Answer
628.2k+ views
Hint: Benzoic acid means COOH group presence. Alkylation followed by further oxidation in the presence of oxidizing agent makes it easier for the conversion of benzene into Benzoic Acid.
Complete step by step answer:
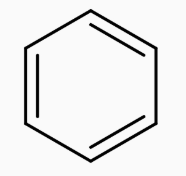
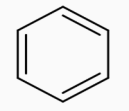
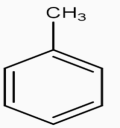
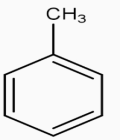
The figure represents the structure of benzene.Our First step is to convert benzene to Toluene. This can be achieved by Alkylation of benzene with $\text{CHC}{{\text{l}}_{3}}.$Along with anhyd $\text{AlC}{{\text{l}}_{\text{3}}}\text{,}$the whole process is called the Friedel Craft Alkylation Process.
$\underset{\text{anhyd}\text{.AlC}{{\text{l}}_{3}}}{\overset{\text{CHC}{{\text{l}}_{3}}}{\mathop{\to }}}\,$
2) Next step is to convert This methyl group into COOH group by the help of strong oxidation agent $\text{KMn}{{\text{O}}_{4}},$the reaction takes place through oxidation.
 $\underset{[\text{oxidation }\!\!]\!\!\text{ }}{\overset{\text{KMn}{{\text{O}}_{4}}}{\mathop{\to }}}\,$
$\underset{[\text{oxidation }\!\!]\!\!\text{ }}{\overset{\text{KMn}{{\text{O}}_{4}}}{\mathop{\to }}}\,$
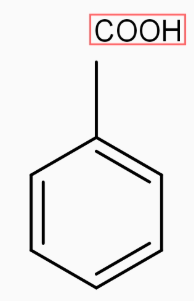 .
.
So, we finally get our final product as Benzoic Acid.
Additional Information: Benzene was first discovered by the English scientist Michael Faraday in 1825 in illuminating gas. In 1834 German Chemist Eilhardt Mitscherlich heated benzoic acid and produced benzene. Benzene is the natural constituent of crude oil and is also one of the most important elements of petrochemical. Due to its cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon.
> Chemical Properties of benzene and benzoic acid:
1) Benzene is immiscible in water but soluble in organic solvents.
2) It is colourless liquid and has an aromatic odour.
3) Benzene shows resonance.
4) It has a density of 0.87g$\text{c}{{\text{m}}^{-3}}$ .
5) Benzoic acid is an aromatic carboxylic acid.
6) Benzoic Acid has a colorless appearance in its solid state, which is of crystalline nature.
Note: We can also convert benzene into benzoic acid by the following steps also:
first convert benzene into benzaldehyde by treating benzene with CO and HCl in the presence of $\text{AlC}{{\text{l}}_{3}}$,And formylation takes place to give benzaldehyde.
Second Step: We can now convert benzaldehyde into benzoic acid in the presence of oxidizing agents. So don't confuse yourself which process should be followed, Both the processes are fine.
Complete step by step answer:

The figure represents the structure of benzene.Our First step is to convert benzene to Toluene. This can be achieved by Alkylation of benzene with $\text{CHC}{{\text{l}}_{3}}.$Along with anhyd $\text{AlC}{{\text{l}}_{\text{3}}}\text{,}$the whole process is called the Friedel Craft Alkylation Process.

$\underset{\text{anhyd}\text{.AlC}{{\text{l}}_{3}}}{\overset{\text{CHC}{{\text{l}}_{3}}}{\mathop{\to }}}\,$

2) Next step is to convert This methyl group into COOH group by the help of strong oxidation agent $\text{KMn}{{\text{O}}_{4}},$the reaction takes place through oxidation.


So, we finally get our final product as Benzoic Acid.
Additional Information: Benzene was first discovered by the English scientist Michael Faraday in 1825 in illuminating gas. In 1834 German Chemist Eilhardt Mitscherlich heated benzoic acid and produced benzene. Benzene is the natural constituent of crude oil and is also one of the most important elements of petrochemical. Due to its cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon.
> Chemical Properties of benzene and benzoic acid:
1) Benzene is immiscible in water but soluble in organic solvents.
2) It is colourless liquid and has an aromatic odour.
3) Benzene shows resonance.
4) It has a density of 0.87g$\text{c}{{\text{m}}^{-3}}$ .
5) Benzoic acid is an aromatic carboxylic acid.
6) Benzoic Acid has a colorless appearance in its solid state, which is of crystalline nature.
Note: We can also convert benzene into benzoic acid by the following steps also:
first convert benzene into benzaldehyde by treating benzene with CO and HCl in the presence of $\text{AlC}{{\text{l}}_{3}}$,And formylation takes place to give benzaldehyde.
Second Step: We can now convert benzaldehyde into benzoic acid in the presence of oxidizing agents. So don't confuse yourself which process should be followed, Both the processes are fine.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




